How the Uruguayan Judiciary Shapes Access to High-Priced Medicines: A Critique through the Right to Health Lens
Lucía Berro Pizzarossa, Katrina Perehudoff, and José Castela Forte
Abstract
Uruguay has witnessed an ever-increasing number of domestic court claims for high-priced medicines despite its comprehensive universal coverage of pharmaceuticals. In response to the current national debate and development of domestic legislation concerning high-priced medicines, we review whether Uruguayan courts adequately interpret the state’s core obligations to provide essential medicines and ensure non-discriminatory access in line with the right to health in the International Covenant on Economic, Social and Cultural Rights. Using a sample of 42 amparo claims for the reimbursement of medicines in 2015, we found that the circuits of appeal fail to offer predictable legal argumentation, including for nearly identical cases. Moreover, the judiciary does not provide an interpretation of state obligations that is consistently aligned with the right to health in the International Covenant on Economic, Social and Cultural Rights. These findings illustrate that medicines litigation in Uruguay offers relief for some individual claims but may exacerbate systemic inequalities by failing to address the structural problems behind high medicines prices. We recommend that the judiciary adopt a consistent standard for assessing state action to realize the right to health within its available resources. Moreover, the legislature should address the need for medicines price control and offer a harmonized interpretation of the right to health. These transformations can increase the transparency and predictability of Uruguay’s health and legal systems for patients and communities.
Introduction
Uruguay has witnessed an ever-increasing number of domestic court claims for high-priced medicines since the dawn of its comprehensive universal health coverage scheme based on the right to health.[1] These cases trigger debates in the courtroom, the media, Parliament, and elsewhere about the scope of the state’s responsibility to provide medicines in an equal and non-discriminatory manner. Uruguay is a party to the International Covenant on Economic, Social and Cultural Rights (ICESCR), which requires that the domestic interpretation and implementation of these rights be consistent with international guidelines. In this case study of Uruguay, we critically examine domestic case law to assess whether and how the judiciary interprets the government’s core obligations under the right to health. The results of our study may assist in current efforts to develop legislation concerning access to expensive medicines in Uruguay. It may also contribute evidence and analysis to the dearth of scholarly debate in the Uruguayan context of health rights litigation.
Uruguay
Uruguay became a democratic republic in 1984 following a period of civil-military rule. With a small but stable population of 3.44 million people, Uruguayans have a longer average life expectancy and lower rate of under-five child mortality than the Latin American and Caribbean regional averages.[2] Uruguay has been a high-income country since 2012, with a gross national income of US$15,230 per capita per year in 2016 (compared to the regional average of US$8,252 per capita). Uruguay’s Human Development Index score increased from 0.692 in 1990 to 0.795 in 2015 (out of 1.0), reflecting improvements to health and life expectancy, access to education and knowledge, and the overall standard of living.[3]
Access to medicines through national health insurance
In 1970, Uruguay ratified the ICESCR and, in doing so, committed to realizing the right to the highest attainable standard of health. In 2007, Uruguay’s National Health Insurance Scheme was introduced as part of a major health system reform grounded in the legal protection of the right to health and access to comprehensive health services.[4] The reform consolidated various insurance financing instruments into the single National Health Fund (Fondo Nacional de Salud), financed by individual and employer contributions, as well as government funds.
The National Medicines Formulary (FTM by its Spanish initials) defines the pharmaceutical benefits package that must be universally available in the health system. It is updated by the Ministry of Public Health based on input from an expert advisory committee that considers the World Health Organization’s Model List of Essential Medicines.[5] The FTM was updated in May 2011, August 2012, January and November 2013, and February 2015.[6]
Domestic legislation enshrines the right to access licensed, quality-assured medicines that are included in the FTM.[7] In practice, access to FTM medicines is granted through two insurance schemes. The first is the National Health Fund, which insures employees, the self-employed, and their families, who have access to the FTM package via a co-payment. The second is the Health Services Administration, which covers the financially vulnerable, who have free-of-charge access to the FTM package.[8] Annex I of the FTM includes the standard pharmaceutical package, while Annex III includes high-cost medicines and other expensive services. Annex III medicines are financed by the National Resource Fund (Fondo Nacional de Recursos), which receives funding from a variety of sources, including the National Health Fund and the Ministry of Economy and Finance.[9] Although 91% of prescribed medicines in Uruguay are generics, spending on high-priced medicines through the National Resource Fund increased from US$2.74 million in 2006 (0.01% of the gross domestic product) to US$19.61 million in 2015 (0.06% of the gross domestic product).[10]
Judicialization of health rights
The judicialization of health is supported by the Uruguayan Constitution, which requires the state to provide the means for prevention and treatment to “indigents” and those lacking sufficient financial resources.[11] The right to health and other fundamental rights—such as the rights to life and to equality and non-discrimination—are justiciable before domestic courts.[12]
Three features of the Uruguayan judicial system contribute to the complexity of health and medicines litigation.
First, the writ of amparo is the judicial mechanism that claimants use in cases where a fundamental constitutional right is at immediate and significant risk. The urgency of the matter warrants an expedited hearing and decision within one week of filing, where the court must render a decision during the hearing (immediately after hearing the respondent’s arguments) or within 24 hours. This expedited proceeding—despite being a key tool to redress alleged human rights violations—restricts the thorough analysis that this topic merits.
Second, the Ministry of Public Health generally appeals decisions against it. Therefore, we assume that most medicines ordered by a court of first instance are ultimately decided by a higher court of appeal.
Third, Uruguay’s civil legal system has seven circuits of appeal and is without binding precedents. Therefore, the result of an appeal will depend on the position adopted by each circuit in each individual case—different from, for example, Argentina’s collective amparo. Each circuit is not bound by previous judgments—not even its own. Moreover, the courts have stated that all cases will be analyzed independently given that, even though they may share certain characteristics, they are not identical.[13]
Fourth, the inconsistency of the decisions of the appellate courts cannot be addressed by a higher court, as the Uruguayan system does not allow for another instance of judicial review for amparo cases. As a result, there is no legal mechanism to require or enforce a harmonized interpretation across courts.
Litigation for expensive medicines
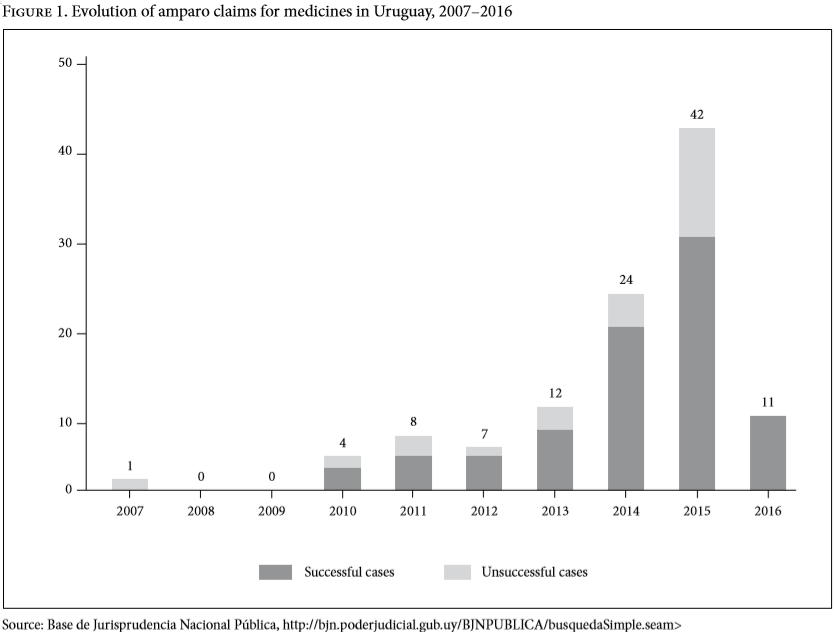
Beginning in 2008, medicines litigation in Uruguay increased steadily, peaking in 2015 (see Figure 1). Such litigation often relates to high-priced medicines included in Annex III of the FTM.[14] The Ministry of Public Health reports that court-ordered expenditure on medicines, which increased 65% between 2010 and 2016, is likely to increase inequities in access to not only high-priced drugs but also basic health services across the population.[15] Moreover, the Ministry of Health spent US$5.3 million providing court-ordered, high-cost medicines in 2017.[16] This unforeseen expenditure was in addition to the 9.2% of gross domestic product that Uruguay already spends on health.[17] This evolution triggered several curious developments in domestic law and policy for pharmaceuticals.
First, the 2015–2019 national budget initially stated that the Uruguayan government would not be “responsible” for medicines and treatments excluded from the FTM.[18] Following much debate, this article was later modified to read that the government would be responsible only for providing medicines of “proven effectiveness.” In this way, the national budget appealed to scientific criteria on “effectiveness” as a measure to discern which medicines the state must provide, rather than leaving the matter to a case-by-case analysis.
Second, Ministerial Order 86/2015 of February 2015 reiterated that an explicit list of pharmaceuticals—including cetuximab, lenalidomide, and sorafenib—were considered cost-ineffective for specific cancers and consequently would not be included in the FTM for these indications.[19] Curiously, decisions granting some of these medicines to plaintiffs continued throughout 2015 and 2016.[20]
Third, the Ministry of Public Health created a new administrative procedure as an alternative to the courts for patients seeking access to off-formulary medicines.[21] This procedure was introduced to stem the number amparo claims; however, it has been criticized as laborious and requiring substantial documentation.[22]
Despite these measures, amparo claims for medicines continued. In 2016, court-ordered medicines consumed 25% of the Ministry of Public Health’s operating expenses.[23] Currently, legislation is being developed to improve access to expensive medicines; however, there is a critical lack of analysis of these amparo claims to inform lawmakers. Specifically, there has been little exploration of whether the courts adequately interpret the state’s core obligations to provide essential medicines and ensure non-discriminatory access in line with the right to health in the ICESCR.
Methods
Using the United Nations Committee on Economic, Social and Cultural Rights’ authoritative interpretation of the right to health in General Comment 14 as its normative framework (described below), our study reviews domestic case law concerning access to medicines. It critically examines how Uruguayan courts determine the scope and boundaries of the state’s action in light of two core obligations under the right to health in the ICESCR.
We selected amparo cases decided in 2015—the year with the most medicines-related decisions since the 2006 health reform. This method offers a snapshot of judicial reasoning at the peak of pharmaceutical claims and in the period coinciding and immediately following a series of legislative changes that were designed to curb amparo cases for medicines.
Cases were retrieved from the online repository of the Uruguayan national judiciary using the keywords “acceso” and “medicamento.” Only cases claiming access to a pharmaceutical intervention through a writ of amparo were included.
We extracted key features of each case into a database for further analysis. These features include the medication and indication requested, the factual and legal basis of the plaintiff’s claim, the legal reasoning of the court, and the decision.
Analytical framework
Our analytical framework is founded on states’ core obligations to realize the right to health, identified in General Comment 14, which was issued in 2000 by the Committee on Economic, Social and Cultural Rights. States’ non-derogable core obligations to realize the right to health form the basic minimum floor of the right on which all other aspects should be built.[24] Core obligations include the duty to provide essential medicines, as defined by the World Health Organization (WHO), and the duty to ensure access to health facilities, goods, and services on a non-discriminatory basis.[25]
First, we examine whether Uruguayan courts have addressed the duty to provide essential medicines in a manner consistent with General Comment 14. Essential medicines are defined by WHO as effective, safe, and comparatively cost-effective to treat the priority health conditions of a population.[26] Every two years, WHO updates its Model List of Essential Medicines, which serves as a guide for domestic governments in their development of local and national lists of essential medicines that respond to local contingencies, such as available public resources and disease burden. In recent years, highly effective and expensive medicines for HIV, hepatitis C infections, and some cancers were added to the WHO model list despite their high price. This move proved that high cost as such does not preclude essentiality; instead, it confirmed the message from WHO’s definition that essential medicines, once selected, must become affordable for all who require them.[27] In the Uruguayan context, the entire FTM (Annexes I and III) is compiled with a comparable objective and according to similar criteria as an essential medicines list. Therefore, the FTM can be considered the national list of essential medicines in the Uruguayan context.
Second, we investigate whether the courts address the core obligation to ensure the right of access to health facilities, goods, and services on a non-discriminatory basis in line with guidance in General Comment 14. According to this general comment, the state has a duty “to prevent any discrimination on internationally prohibited grounds in the provision of health care and health services, especially with respect to the core obligations of the right to health.”[28] It warns against “inappropriate health resource allocation” that may lead to discrete discrimination. It offers the example of favoring “expensive curative health services which are often accessible only to a small, privileged fraction of the population, rather than primary and preventive health care benefiting a far larger part of the population.”[29] Moreover, it notes that the state has a “special obligation” to provide health insurance and care to those with insufficient means.[30] This duty is closely related to the universal entitlement to “a system of health protection which provides equality of opportunity” such that people can enjoy their health rights.[31]
Results
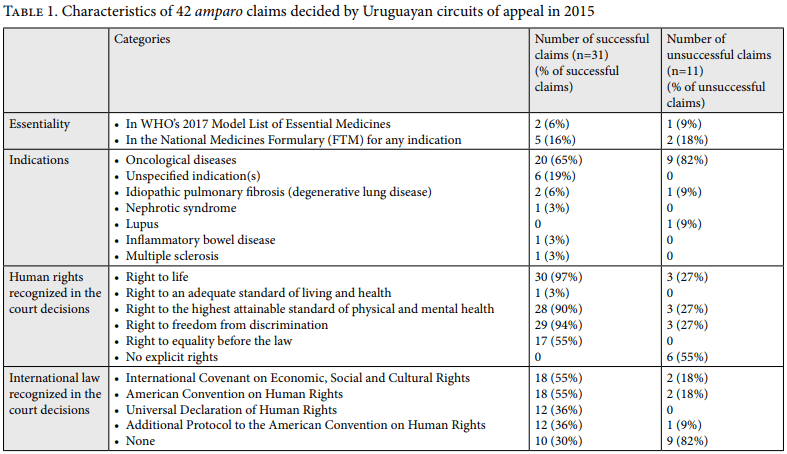
Of the 52 claims decided in 2015 that were available in the judicial repository, 10 were excluded (3 claimed medical devices as opposed to pharmaceuticals, and 7 were not amparo cases), leaving 42 claims that were included in this study (see Table 1). Each of these claims sought one pharmaceutical. Of the 42 claims, 31 (74%) were decided in favor of the plaintiff (hereafter “successful claims”).
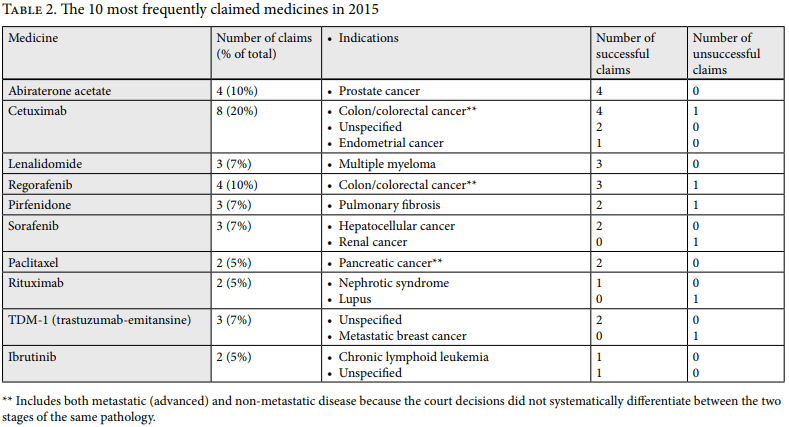
Thirty-four claims (81%) accounted for 10 medicines (see Table 2). Requests were most frequently for licensed medicines not included in the FTM (hereafter “off-formulary”). Eight claims (19%) successfully acquired the off-formulary medicines cetuximab, lenalidomide, and sorafenib for cost-ineffective indications that were explicitly excluded from the FTM by Ministerial Order 86/2015.[32] The courts denied two claims for unlicensed medicines and accepted the only request for an on-formulary medicine.
Duty to provide essential medicines
In cases requesting off-formulary medicines, the courts produced vague and sometimes contradictory evaluations of alleged violations of the fundamental right to health. For example, one court granted reimbursement of off-formulary medicines despite “bureaucratic reasons” for not adding the medicines to the list (for example, passage of the annual submission deadline to the advisory committee or insufficient time to complete the technical appraisal of a medicine for inclusion). However, in three other cases, the courts reasoned that an ongoing assessment or insufficient time to evaluate cost-effectiveness justified not reimbursing the medicine at that time. The latter three decisions are consistent with the core obligation to provide essential medicines, which presupposes that sufficient information and time has been given to adequately assess the essentiality of each medicine—an imperative step for inclusion in the FTM.
Similar inconsistencies are evident in decisions not to reimburse a high-priced medicine in light of limited public funds, which the courts interpreted as a breach of fundamental rights in six cases. However, in four other cases, the courts reasoned that not reimbursing expensive medicines due to limited resources or their lack of cost-effectiveness was consistent with fundamental rights. The former decisions are consistent with the concept of non-derogable core obligations in General Comment 14. The four latter cases reflect a softer approach to core obligations, which is addressed further in the Discussion section below.
Two similar claims for cetuximab demonstrate this inconsistent reasoning. Cetuximab’s reported price for colon cancer is US$190,483 per patient per year.[33] Excluded from the FTM due to its cost-ineffectiveness, cetuximab was claimed twice in our 2015 sample for the treatment of metastatic colon cancer. In the first case, decided on October 10, 2015, Circuit 7 determined that there was no scientific justification for excluding cetuximab for this indication. In reaching this conclusion, the court reasoned that a lack of cost-effectiveness did not justify denying reimbursement to a patient who could not otherwise afford it.[34] However, in the second case, decided on November 3, 2015, Circuit 5 decided that it must respect the decision to omit cetuximab for this indication from the FTM on economic grounds. In this court’s appreciation, this decision was consistent with the patient’s fundamental rights and previous court rulings.[35]
Duty to ensure non-discriminatory access to health care
Uruguayan courts conceptualize equality and non-discrimination in two different ways, leading to two significantly different results.
On one hand, successful cases have generally found a breach of the principle of equality and non-discrimination on two grounds. First, some courts consider that the positive market authorization decision and negative reimbursement decision (in other words, exclusion from the FTM) regarding certain high-priced medicines breaches the right to equality and non-discrimination. This is because authorization without reimbursement allows access for those who can afford the medicines but not for those without the financial means and who are limited to the FTM selection. The reasoning is based on the idea that “every patient has the right to access medicines of quality, and the constitutional protection of this right does not distinguish whether these medicines are or are not included in the FTM.”[36] In the words of Circuit 7 “The effective protection of the right to life or health cannot depend … on one’s financial ability or privileged situation that enables them to access the medical treatment.”[37] According to this line of decisions, “economic accessibility” shall be guaranteed by the state through the provision of all medicines, irrespective of their cost or their inclusion in the FTM.[38] The courts’ notion of providing for those who cannot provide for themselves appears to align with the right to health’s concept of equality of opportunity. However, the state is not obliged to provide immediate access to health services of any cost to those dependent on state health care. Moreover, General Comment 14 cautions states against discrimination that can result from providing expensive curative care to the few at the expense of preventative and primary care for the many.
Second, courts have generally been receptive to the argument that since both the Health Services Administration (health insurance for the financially vulnerable) and the Ministry of Public Health may have provided a particular medicine to other patients who have requested it previously, not granting a plaintiff’s request would breach the principle of equality enshrined in article 8 of the Uruguayan Constitution. This is because other patients with similar conditions have been granted access to the medicine in question (either by judicial or administrative action). The courts consider that the Ministry of Public Health decides these issues in an arbitrary and discriminatory manner that breaches the right to equality. They conclude that the use of public resources cannot discriminate between citizens and that the Ministry of Public Health does not have a valid reason to justify the difference in treatment.
On the other hand, the courts have also used the argument of equality in their decisions not to grant a plaintiff’s request. These decisions sustain that the government has limited resources to attend to the health care needs of the whole population and that the provision of certain high-priced medicines can clash with the needs of the rest of the population. For example, Circuit 6 has argued that “the primary obligation of the Ministry of Public Health is to attend to the general welfare applying the principle of equality, not just for one patient but for everybody.”[39] Along the same lines, Circuit 5 has viewed plaintiffs’ requests as a demand for special treatment “at a high cost and over the needs of the rest of the population.”[40] This reasoning is somewhat consistent with the concept of “inappropriate health resource allocation” in General Comment 14. Circuit 5 has pointed out that “even when the condition of the patient is grave—unfortunately—this is not the only person that needs to be assisted … That is the key issue here.”[41] This argument points to the fact that increased judicialization distorts health planning and priority setting, forcing decisions that reflect on the individual cases being judged and not on society’s collective needs. The courts emphasize the fact that decisions regarding health policies—which require the consideration of multiple factors—should be made by the executive branch. Judicial intervention to grant access to high-priced medicines—without a grave cause to justify it—can endanger the general well-being of the population by distorting the national health budget. According to this approach, “[j]udges need to be guided by the law and what is just, not only for the plaintiff but for others and society as a whole.”[42] It has been argued that deciding otherwise will turn the courts into a “judicial pharmacy.”[43]
Discussion
The majority of the claims in our sample concerned one of ten off-formulary medicines frequently requested to treat cancer. The courts provided inconsistent and unreliable legal reasoning in their decisions for the protection of the right to health. Our study shows that the courts’ reasoning neither implicitly nor explicitly engages with the concept of core obligations to provide essential medicines in a non-discriminatory manner. Although some decisions are consistent with the Committee on Economic, Social and Cultural Rights’ interpretation of core obligations, we cannot determine whether these were conscious or coincidental judicial rulings.
These findings illustrate that Uruguayan case law from 2015 fails to provide any legal certainty regarding the boundaries of the state’s core obligation to provide essential medicines in a non-discriminatory manner. Due to a lack of consistency, these decisions may further exacerbate, rather than remedy, inequalities among patients with comparable health needs and within the publicly funded pharmaceutical reimbursement system as a whole.[44]
A softer approach to core obligations in international human rights law
A more flexible approach to minimum core obligations seems to be condoned in the 2013 Optional Protocol to the ICESCR (hereafter ICESCR-OP), inspired by the 2000 Grootboom and 2001 Treatment Action Campaign decisions of the South African Constitutional Court.[45] ICESCR-OP is the first instrument to enable the international enforcement of the rights laid out in the ICESCR. It adopts a standard of reasonableness suggesting that social rights realization is contingent on an assessment of whether the state has taken sufficiently appropriate measures to realize the right within its maximum resource limits. As Bruce Porter explains:
Reasonableness is a contextual inquiry into the content of Covenant rights in particular circumstances, attending equally to both the voice and experiences of claimants, and to the realities, restraints, and difficult choices faced by governments. What is reasonable will depend as much on the nature of the interest at stake and the unique circumstances of the particular claimant or group, as on budgetary constraints, competing needs and policy rationale presented by the state.[46]
By ratifying the ICESCR-OP in 2013, Uruguay expressly agreed to be held accountable before an international committee to the instrument’s standards and principles, such as the nascent concept of reasonableness.
The standard of reasonableness: A measure of state action for the Uruguayan judiciary?
Our results show that despite considering the contextual needs and restraints of the plaintiff and the state in each decision, the Uruguayan judiciary has not applied a common measure to judge state action. One of the present authors (Katrina Perehudoff) and Lisa Forman propose that the standard of reasonableness, found in South African jurisprudence and the ICESCR-OP, may serve as a lens through which we can interpret core obligations. In other words, the standard of reasonableness can help give substance to the state’s duty to use all available resources to satisfy its core obligations toward essential medicines.[47] In particular, Perehudoff and Forman suggest that satisfying core obligations in the context of available resources can be delineated into four duties: (1) ensure sufficient government spending on pharmaceuticals, (2) ensure efficient spending on pharmaceuticals, (3) generate efficiencies by seeking international cooperation and assistance, and (4) observe non-discrimination in pharmaceutical policy.[48] Uruguayan lawmakers could be expected to align the domestic interpretation and enforcement of social rights with the international standards to which the Uruguayan state has agreed to be accountable.
In the case of Uruguay, we assert that the judiciary could seek inspiration from the standard of reasonableness to assess claims for high-priced medicines. An examination of core obligations consistent with the standard of reasonableness would assess whether the state had taken all “reasonable” measures to provide the medicine before determining whether the state violated rights.
Let us take a look at the three claims for lenalidomide identified in our study in order to illustrate an alternate line of judicial assessment inspired by the standard of reasonableness. The Ministry of Public Health determined in 2013 that lenalidomide is cost-ineffective for the second-line treatment of multiple myeloma and, consequently, did not include the medicine in the FTM. However, the health technology assessment notes that a 70% price reduction would render lenalidomide sufficiently cost-effective for FTM inclusion.[49] In 2015, we found that three patients who claimed lenalidomide were granted court-ordered reimbursement for multiple myeloma despite it being off-formulary for this indication. In each of these cases, the courts determined that failing to reimburse a high-priced medicine that is proven effective for a life-threatening condition on the grounds of limited state resources violates the rights to life, health, and non-discrimination.[50]
Uruguayan scholars note that in response to high-priced medicines, price regulation and international cooperation for joint purchasing and price transparency is being pursued in Uruguay.[51] Therefore, the courts could have considered whether similar measures were pursued in relation to lenalidomide prior to deciding in the plaintiffs’ favor. Recalling the four duties proposed by Perehudoff and Forman, this line of reasoning could have examined whether the state took measures to maximize its public pharmaceutical budget (duty 1) and spend efficiently (duty 2), such as through the use of price controls and TRIPS flexibilities when all other measures fail to yield affordable medicines.[52] Sufficient and efficient spending can mitigate the need for discriminatory trade-offs and care rationing (duty 4).[53] Finally, the court could have questioned whether the state took steps to jointly procure medicines with larger neighboring countries (duty 3) in order to leverage economies of scale. By examining the “reasonableness” of state efforts to fulfill Uruguay’s core obligations, the judiciary could have secured more equitable access to lenalidomide for the plaintiffs while also triggering important policy changes that would grant access to the other invisible patients with multiple myeloma who did not file a writ of amparo, while still respecting the separation of powers.
Does litigation stimulate rather than remedy health inequality?
This “wave” of litigation since Uruguay’s health reform is likely to have affected equity in the country’s tax-funded universal health system in several ways. First, not all consumers with unmet health needs are equally able to access a court. This concern is corroborated by government representatives who claim that health rights litigation may result in preferential access for people of higher socioeconomic status.[54] Second, successful plaintiffs inevitably receive and consume more health system resources than those who do not seek treatment through the courts.[55] Third, these challenges are compounded by inconsistent judicial outcomes in highly similar cases.
Uruguayan case law in our sample provided little information about the plaintiffs’ socioeconomic status. Therefore, we are unable to assess whether litigation is exacerbating inequalities by providing preferential reimbursement of medicines to the better-off, as has been reported elsewhere in the region.[56] However, the question of inequality warrants further research in Uruguay considering the above factors at play.
Access to the courts for people who cannot afford a lawyer is stimulated through several initiatives of the law clinics of the Universidad de la República (a public university). However, securing representation by these clinics is limited by their case load and by patients’ ability to travel to the capital city in order to access the clinics’ services. While the clinics’ work may palliate inequalities in access to the courts, unless all health consumers have the same political and economic resources, certain groups are more likely to be able to litigate—and therefore access high-priced drugs—more effectively than others. Considering that socioeconomic status is not only one of the most prominent social determinants of health but also an important indicator of one’s ability to access the courts, we must agree with Octavio Ferraz that “the ability to access the judiciary is not a fair criterion for the allocation of health resources.”[57]
Domestic policy recommendations
Two key policy recommendations arise from these findings. First, the Uruguayan state should consider legislative measures to control the prices of expensive medicines that would otherwise be eligible for inclusion in the FTM if it were not for their prohibitive price. In this line, a bill is currently being debated in Parliament to provide tax cuts to private companies that donate to the National Resource Fund, which finances expensive medicines.[58] At the time of writing, it is unclear whether the bill will be adopted. In July 2017, the Committee on Economic, Social and Cultural Rights urged the country to accelerate the passing of this bill in order to guarantee access to all medicines needed to enjoy the right to health.[59] While the bill is an attempt to expand the available budget for and access to expensive medicines, it does not address the underlying reasons for high prices and the use of all available means of reducing prices as recommended by the Lancet’s Commission on Essential Medicines Policies.[60] This fragmented approach gives in to the lobbying power of pharmaceutical companies that will benefit from tax cuts for donating money to the National Resource Fund while having their high-priced medicines be included in the FTM. In principle, donations are not recommended as a means for improving access to high-priced products, as they allow pharmaceutical companies to maintain the underlying high prices.
We recommend that if the political climate allows for a legislative solution, Uruguay should pursue a more holistic law that regulates and supports all possible price control measures and promotes international cooperation to evaluate and purchase medicines. Moreover, a more transparent and participatory process to establish the criteria for inclusion in the FTM would also foster open public debate and a deeper public understanding of the issues at play.[61] Technical decisions about whether to include new medicines in the FTM should be objective, consistent, and evidence based. Failing to take measures such as these to make essential medicines affordable to all is inconsistent with the right to health.[62]
Second, alternative approaches to the writ of amparo are needed. Uruguay should also allow for another instance of judicial review that harmonizes the inconsistent decisions on appeal. Such an alternative approach should ensure that all courts of appeal interpret the law in a uniform manner, which would help reduce disparate judicial outcomes in highly similar cases. Alternative approaches may be found in neighboring countries that also face numerous judicial claims for medicines.
Study limitations
The limitations of our study relate to the accessibility and completeness of data in court decisions. First, our search of the official online repository produced only 52 decisions from 2015, while Uruguayan scholars report retrieving 80 such decisions for the same year.[63] We consulted one of these scholars, and although both of our research teams reported inconsistencies in the repository’s search function, neither team could identify a solution. We could not access a list of the 80 medicines decisions, and time restraints precluded a manual search of the repository. Therefore, we proceeded with this convenience sample of 52 decisions. Although we cannot claim that our sample is representative of all medicines claims from 2015, it does represent the decisions that are most readily accessible to the Uruguayan judiciary, which uses the same online repository to access case law. We hypothesize that judges and their teams are most likely to consult case law that is the easiest to access, especially considering that the courts of appeal hear and decide amparo claims for medicines within one week of filing. Moreover, selection bias favoring claims for high-cost, off-formulary medicines in our sample is unlikely because the Ministry of Public Health tends to appeal all decisions against it. This means that if a court of first instance ordered the ministry to reimburse an on-formulary medicine, then the government would appeal the decision, which would then be heard by a court of appeal and therefore appear in our sample.
Second, several court decisions contained little to no information about the pathologies that the medicine in question was requested to treat (see the five cases with an “unspecified” indication in Table 2). As a result, our finding that courts rule inconsistently on the same indication may be more frequent than we documented.
Third, the fact that General Comment 14 is not binding on states could call into question its legitimacy as an analytical framework. Nevertheless, this general comment is an authoritative interpretation of the right to health in the ICESCR. It instructs state parties on their goals and actions required to attain the right to health for all; it also reflects the monitoring criteria applied by the Committee on Economic, Social and Cultural Rights. General Comment 14 is also instructive for domestic law and policymaking, being explicitly and implicitly referenced in national medicines policies, domestic health legislation, and medicines case law from various jurisdictions.[64] From these examples, we can conclude that General Comment 14 is the most authoritative human rights guide for domestic health law and policymaking despite the fact that it does not reflect all aspects of a public health or health systems approach.
Conclusion
Our findings show that Uruguayan case law concerning high-priced medicines fails to offer predictable legal argumentation among the country’s seven circuits of appeal. Nor does this body of case law provide an interpretation of state obligations that is consistently aligned with the right to health in the ICESCR. While medicines litigation in Uruguay offers relief for some individual claims, the courts’ inconsistent legal reasoning has the potential to exacerbate systemic inequalities by failing to address the structural problems behind high medicines prices. In response, future court rulings should embrace a consistent standard for examining state action to realize the right to health within its available resources. Furthermore, future legislative responses should address the need for medicines price control and offer a harmonized interpretation of these rights and obligations. These steps will increase the transparency and predictability of Uruguay’s health and legal systems for patients.
Acknowledgments
We are indebted to Hans Hogerzeil and Marlies Hesselman for their thoughtful comments on an earlier version of this article.
Lucía Berro Pizzarossa, MJur, is a doctoral candidate at the International Law Department and Global Health Law Groningen Research Centre at the University of Groningen, the Netherlands, and a legal advisor for MYSU (Women and Health) in Uruguay.
Katrina Perehudoff, MSc, LLM, is a doctoral candidate in the Global Health Unit, Department of Health Sciences in the University Medical Centre, University of Groningen, the Netherlands.
José Castela Forte, BSc, is a medical student, student researcher in Anesthesiology and Intensive Care, and student assistant in the Global Health Unit, Department of Health Sciences in the University Medical Centre, University of Groningen, the Netherlands.
Please address correspondence to Katrina Perehudoff. Email: katrina.perehudoff@gmail.com.
Competing interests: None declared.
Copyright © 2018 Berro Pizzarossa, Perehudoff, and Castela Forte. This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
References
[1]. F. Borgia, “Health in Uruguay: Progress and challenges in the right to health care three years after the first progressive government,” Social Medicine 3 (2008).
[2]. World Bank, Uruguay (2015). Available at https://data.worldbank.org/country/uruguay.
[3]. United Nations Development Programme, Briefing note for countries on the 2016 Human Development Report (2016). Available at http://hdr.undp.org/sites/all/themes/hdr_theme/country-notes/URY.pdf
[4]. Law No. 18211 of 2007 on the creation, functioning and financing of the National Integrated Health System, art. 1.
[5]. Decree 265/006 of 2006 on the creation of the National Medicines Formulary.
[6]. Decision 108/2015, Court of Appeals 1st Circuit.
[7]. Law No. 18335 of 2008 on the rights and duties of patients and users of health care services. Available at https://legislativo.parlamento.gub.uy/temporales/leytemp7610153.htm.
[8]. Ibid., art. 7.
[9]. Pan American Health Organization, “Access to high-cost medicines in the Americas: Situation, challenges, and perspectives,” Technical Series No. 1 (Washington, DC: Pan American Health Organization, 2010).
[10]. G. Bardazano, V. Caredio, P. Cechi, et al., Múltiples miradas a los medicamentos de alto costo: hacia una comprensión integral del tema (Montevideo: Universidad de la República, 2017), p. 86. Available at http://www.proyectomac.hc.edu.uy/images/múltiples_miradas_a_los_medicamentos_de_alto_costo-_hacia_una_comprensión_integral_del_tema.pdf; G. Lezama and P. Triunfo, Medicación de alto costo, Documento de Trabajo 04/16 (Montevideo: Universidad de la República, 2016).
[11]. Uruguayan Constitution (1966, last amended 2004), sec. 44.
[12]. Ibid., sec. 8.
[13]. Decision 411/2015, Court of Appeals 5th Circuit.
[14]. Lezama and Triunfo (see note 10).
[15]. “Fármacos caros son el 25% del presupuesto del MSP,” El Observador (June 23, 2017). Available at http://www.elobservador.com.uy/farmacos-caros-son-el-25-del-presupuesto-del-msp-n1088306.
[16]. “Uruguay Ministerio de Salud gasto mas de us 5 millones en medicamentos caros,” El Observador (March 16, 2018). Available at https://clustersalud.americaeconomia.com/farmaceuticas/uruguay-ministerio-de-salud-gasto-mas-de-us-5-millones-en-medicamentos-caros.
[17]. Ibid.
[18]. “Medicamentos de alto costo: van 40 juicios de los que ya se ganaron 31,” Ecos Uruguay (July 18, 2017). Available at http://ecos.la/UY/5/salud/2017/07/18/15359/medicamentos-de-alto-costo-van-40-juicios-de-los-que-ya-se-ganaron-31.
[19]. Ordinance 86/2015 of February 27, 2015. Available at ww.msp.gub.uy/sites/default/files/archivos_adjuntos/Ordenanza%20N°%2086.pdf.
[20]. “Medicamentos de alto costo” (see note 18).
[21]. Ordinance 882/2015 of December 9, 2015. Available at http://www.shu.com.uy/images_hematologia/descargas/ordenanza-882-msp.pdf; Ordinance 692/2016 of August 2016. Available at http://www.msp.gub.uy/sites/default/files/archivos_adjuntos/ordenanza%20692-2016.pdf.
[22]. Ordinance 692/2016 (see note 21); Bardazano et al (see note 10), pp. 81–83.
[23]. “Rendición de cuentas a estudio del Parlamento,” El Observador (July 7, 2017). Available at http://www.elobservador.com.uy/proponen-negociar-mejorar-acceso-medicamentos-n1094292.
[24]. Committee on Economic, Social and Cultural Rights, General Comment No. 14, The Right to the Highest Attainable Standard of Health, UN Doc. E/C.12/2000/4 (2000), paras. 43, 47; B. Griffey, “The ‘reasonableness’ test: Assessing violations of state obligations under the Optional Protocol to the International Covenant on Economic, Social and Cultural Rights,” Human Rights Law Review 11/2 (2011), pp. 275–327; L. Forman, “Can minimum core obligations survive a reasonableness standard of review under the Optional Protocol to the International Covenant on Economic, Social and Cultural Rights?,” Ottawa Law Review 47 (2015), p. 561.
[25]. Committee on Economic, Social and Cultural Rights (2000, see note 24), para. 47.
[26]. World Health Organization, “The selection and use of essential medicines,” WHO Technical Report Series No. 914 (Geneva: WHO, 2003).
[27]. A. Gray, V. Wirtz, E. ‘t Hoen, et al., “Essential medicines are still essential,” Lancet 386/10004 (2015), pp. 1601–1603.
[28]. Committee on Economic, Social and Cultural Rights (2000, see note 24), para. 19.
[29]. Ibid.
[30]. Ibid.
[31]. Ibid., para. 8.
[32]. Ordinance 86/2015 (see note 19).
[33]. A. Croci, A. German, R. Alonso, et al., Evaluation of cost-utility of using cetuximab last line treatment of metastatic colorectal cancer (Health Assessment Division, Directorate General of Health, Ministry of Public Health, 2014); Bardazano et al. (see note 10), p. 80.
[34]. Decision 154/2015, Court of Appeals 7th Circuit.
[35]. Decision 572/2015, Court of Appeals 5th Circuit.
[36]. Decision 154/2015, Court of Appeals 7th Circuit.
[37]. Decision 103/2015, Court of Appeals 7th Circuit.
[38]. Decision 129/2015, Court of Appeals 2nd Circuit.
[39]. Decision 105/2015, Court of Appeals 6th Circuit.
[40]. Decision 150/2015, Court of Appeals 5th Circuit.
[41]. Ibid.
[42]. Ibid.
[43]. Ibid.
[44]. A. Aleman and A. Galan, “Impact of health technology assessment in litigation concerning access to high-cost drugs,” International Journal of Technology Assessment in Health Care (2017) pp. 1–4; A. P. Barcellos, “Sanitation rights, public law litigation, and inequality: A case study from Brazil,” Health and Human Rights Journal 16/2 (2014), p. 36.
[45]. L. Forman, “What future for the minimum core?,” in J. Harrington and M. Stuttaford (eds), Global health and human rights: Legal and philosophical perspectives (New York, NY: Routledge, 2010), pp. 71–73.
[46]. B. Porter, “Reasonableness and Article 8(4),” in M. Langford, B. Porter, R. Brown, and J. Rossi (eds), Optional Protocol to the International Covenant on Economic, Social and Cultural Right: A commentary (Pretoria: Pretoria University Press, 2016), p. 217.
[47]. Committee on Economic, Social and Cultural Rights, An evaluation of the obligation to take steps to the “maximum of available resources” under an Optional Protocol to the Covenant, UN Doc. E/C.12/2007/1 (2007).
[48]. Ibid.
[49]. E. Villamil, A. German, A. Perez, and A. Galan, “Evaluation cost-utility of using lenalidomida for treating multiple myeloma (2nd line)” (Group Health Technology Assessment Evaluation Division, Directorate General of Health, Ministry of Public Health, 2013); Bardazano et al. (see note 10), p. 80.
[50]. Decision 103/2015, Court of Appeals 7th Circuit; Decision 002/2015, Court of Appeals 2nd Circuit; Decision 128/2015, Court of Appeals 7th Circuit.
[51]. Bardazano et al. (see note 10), pp. 80, 116.
[52]. K. Perehudoff and L. Forman, “What constitutes ‘reasonable’ state action on core obligations? Considering a right to health framework to provide essential medicines” (November 2017), on file with the authors.
[53]. J. Kutzin, “Health financing for universal coverage and health system performance: Concepts and implications for policy,” Bulletin of the World Health Organization 91/8 (2013), pp. 602–611.
[54]. Aleman and Galan (see note 44).
[55]. “Los problemas para acceder a medicamentos de alto costo en Uruguay,” 970AM Universal (April 10, 2017). Available at https://970universal.com/2017/04/10/los-problemas-acceder-medicamentos-alto-costo-uruguay.
[56]. J. Biehl, M. P. Socal, and J. J. Amon, “The judicialization of health and the quest for state accountability: Evidence from 1,262 lawsuits for access to medicines in southern Brazil,” Health and Human Rights Journal 18/1 (2016), pp. 209–220; A. Yamin and O. Parra-Vera, “How do courts set health policy? The case of the Colombian Constitutional Court,” PLoS Medicine 6/2 (2009), p. e1000032.
[57]. O. L. Motta Ferraz, “Health inequalities, rights and courts: The social impact of the judicialization of health,” in A. Yamin and S. Gloppen (eds), Litigating health rights: Can courts bring more justice to health? (Boston: Harvard University Press, 2011), pp. 99–100.
[58]. Comisión de Salud Publica, carpeta 1698, year 2016, High Cost Medicines.
[59]. Committee on Economic, Social and Cultural Rights, Concluding Observations on Uruguay, UN Doc. E/C.12/URY/CO/5 (2017), paras. 46–47.
[60]. V. Wirtz, H. Hogerzeil, A. Gray, et al., “Essential medicines for universal health coverage,” Lancet 389/10067 (2017), p. 21.
[61]. K. Perehudoff, B. Toebes, and H. Hogerzeil, “A human rights-based approach to the reimbursement of expensive medicines,” Bulletin of the World Health Organization 94/12 (2016), pp. 935–936.
[62]. Ibid.
[63]. Bardazano et al. (see note 10).
[64]. Consejo Nacional de Política Económica y Social (Colombia), Política Farmacéutica Nacional [National Pharmaceutical Policy] (August 2012), pp. 6–7; Colombian Ministry of Health, Law No. 1751 of 2015. Available at https://www.minsalud.gov.co/Normatividad_Nuevo/ Ley%201751%20de%202015.pdf; Mathew Okwanda v. Minister of Health & Medical Services and others (High Court of Kenya), Petition No. 94 of 2012.

