Leslie London, Helen Cox, and Fons Coomans
Abstract
The right to enjoy the benefits of scientific progress (REBSP) is a little-known but potentially valuable right that can contribute to rights-based approaches to addressing multidrug-resistant TB (MDR-TB). We argue that better understanding of the REBSP may help to advance legal and civil society action for health rights. While the REBSP does not provide an individual entitlement to have a new drug developed for MDR-TB, it sets up entitlements to expect a state to establish a legislative and policy framework aimed at developing scientific capacity to address the most important health issues and at disseminating the outcomes of scientific research. By making scientific findings available and accessible, people can be enabled to claim the use of science for social benefits. Inasmuch as the market fails to address neglected diseases such as MDR-TB, the REBSP provides a potential counterbalance to frame a positive obligation on states to both marshal their own resources and to coordinate the actions of multiple other actors towards this goal, including non-state actors. While the latter do not hold the same level of accountability as states, the REBSP can still enable the recognition of obligations at a level of “soft law” responsibilities.
Introduction
In this article, we explore the potential of the right to enjoy the benefits of scientific progress and its applications (REBSP), a right included in Article 27 of the Universal Declaration of Human Rights (UDHR) and Article 15 of the International Covenant on Economic, Social and Cultural Rights (ICESCR), as an important lever for a rights-based approach to improving treatment for neglected diseases. We use the example of multidrug-resistant tuberculosis (MDR-TB), a disease with disproportionate impacts on the poor and marginalized, to highlight relevant questions for further discussion.
Better understanding of the REBSP may well facilitate greater likelihood that patients and communities will realize their rights to health, and greater clarity on obligations of state and non-state actors in responding to the crisis that is MDR-TB in the world today. While the REBSP is slowly receiving increased attention from UN human rights bodies and academics, it remains largely overlooked, so greater attention to this right, in relation to the right to health, is timely.1 Today’s world is increasingly turning to science and technology for solutions to persistent socioeconomic and development problems, and the human dimension of science therefore demands increased attention.
MDR-TB is both a major global health challenge and a clear illustration of how the REBSP might interface with the right to health.2 About half a million new cases are estimated to occur each year, with the majority the result of direct transmission.3 Yet only one patient in five has access to currently recommended treatment, and only half of these patients are cured effectively with that treatment.4 The reasons for lack of treatment are multifactorial and include poor access to MDR-TB diagnosis, lengthy and complicated treatment regimens, and the high cost of drugs and poor effectiveness of the regimen itself.5 Currently recommended regimens for MDR-TB consist of older drugs previously discarded for first-line TB treatment, and “repurposed” drugs, that is, drugs developed for indications other than TB. These drugs are combined in multi-drug regimens based primarily on expert advice and limited observational data, and result in cure for 60% of patients even under ideal conditions.6 Clearly a more effective and more accessible treatment regimen for MDR-TB is required, both to cure individuals and to prevent ongoing community transmission of this airborne infectious disease. Human rights barriers to effective TB treatment include discrimination and exclusion of sick people, often in combination with poverty.7
Could an approach based on the right to enjoy the benefits of scientific progress change that situation? We explore in this article how the REBSP might address a number of relevant questions. Can it be leveraged to facilitate access to existing or repurposed medicines, that is, the existing products of scientific progress? Could it help to spur the development of novel medicines not yet existent in the drug pipeline?
Given that the development of a new medicine is based on scientific research, who has obligations to engage in scientific research? Following development of a new medicine, who has the responsibility to ensure that the new medicine is trialled appropriately and then made accessible to those who need it most? Below we map the origins and contours of the REBSP and its relationship to the right to health; then we review the current burden of MDR-TB and status of MDR-TB drug treatment; we describe actions needed to improve access to cure for MDR-TB; we explore responsibilities for implementation of the REBSP and the role of legal/political action in ensuring human rights; and then we close by proposing future steps to test whether the REBSP can be drawn upon to improve availability of and access to new medicines and thereby improved treatment for MDR-TB patients. While the focus of this paper is on treatment for MDR-TB, we recognize that similar arguments may be made with respect to other aspects of MDR-TB management, including diagnostics, and, indeed, to many other conditions.
Situating the right to enjoy the benefits of scientific progress as a human right
The right to enjoy the benefits of scientific progress as a human right has remained rather obscure and unexplored in human rights discourse. An early commentator argued in favor of establishing a duty for the community to make funding available for scientific research, “as a means for alleviating wants of mankind, for the development of mankind, and for the pursuit of truth.”8 Besides inclusion in Article 27.1 of the UDHR and Article 15.1b of the ICESCR, it is also part of Article 15 of UNESCO’s Universal Declaration on Bioethics and Human Rights. In the period of the drafting of the UDHR and the ICESCR between 1946 and 1966, there was much optimism about the potential contribution of science to improving human life and living conditions of people. At the same time, there was awareness and concern about the collusion of science in the Nazi genocide during World War II. As a result, these human rights instruments explicitly stated that people should benefit from scientific progress.9 An important issue in this respect is whether the REBSP only refers to sharing the concrete benefits of scientific progress among people or whether it also includes the development of science as such. This is crucial from the perspective of investing in scientific research for the benefit of the health situation of poor people, which is less profitable for the corporate sector. The present authors agree with the latter view as advocated by Chapman, because it does justice to one of the key features of human rights: non-discriminatory and equal access to benefits.10
Science as a concept relates to obtaining and expanding knowledge and understanding processes and phenomena which occur in nature and society.11 The link with the realization of human rights can be found in the terms ‘progress’ and ‘benefits’ in Articles 27 and 15. The term ‘progress’ recognizes that science develops in certain ways, sometimes linear and sequential, and sometimes through qualitative shifts, towards creating positive effects for human beings, society at large, and life on earth in general. What was the extent of scientific knowledge decades ago is recognized as being superseded by newer advances in knowledge generated from ongoing scientific research—hence the idea of ‘progress.’ The term ‘benefits’ has a qualitative connotation and includes effects that positively impact on humans and society. Both terms therefore link to the notion of progressive realization of economic, social, and cultural rights as laid out in Article 2.1 of the ICESCR in the sense that progress of science that generates benefits allows for the achievement of higher and more advanced levels of welfare and wellbeing aimed at the full realization of rights over time.12 The REBSP, especially the notions of science and progress, should therefore be seen as a vehicle for achieving other rights, such as the right to health, rather than an end in itself.
Although the REBSP has been phrased as an individual right, we submit that, similar to the right to health, it has a collective dimension. Indeed, it is almost irrevocably social in its expression, rather than individualized.13 For example, in discussing how remedies and accountability operate under the right to health, the General Comment notes that:
Regardless of whether groups as such can seek remedies as distinct holders of rights, state parties are bound by both the collective and individual dimensions of article 12. Collective rights are critical in the field of health; modern public health policy relies heavily on prevention and promotion which are approaches directed primarily to groups.14
This means that, in the same way, enjoying scientific progress will not benefit one person only, or a limited group of people. Rather, it will benefit larger groups of people, or even society at large. This will particularly apply when progress in pharmaceutical research leads to greater availability and accessibility of drugs for the treatment of MDR-TB. A human rights approach which focuses on strengthening access for the most vulnerable and marginalized groups may contribute to achieving equity and thus give clear expression to the collective dimension of the right.
There are clear links with the right to the highest attainable standard of health (ICESCR, Article 12) which includes the right to have access to essential drugs for the treatment of life-threatening diseases.15 This means that existing drugs need to be made accessible without prohibitive cost barriers to those in need. The REBSP can potentially take this further and suggest that essential drugs need to be ‘created’ through scientific research and development in addition to being made accessible. Given this, there is likely to be an underlying tension between, on the one hand, the REBSP and, on the other hand, the protection of intellectual property rights (patents) of inventors and producers (companies and private research institutes).16 For example, exclusivity of pharmaceutical test data, as provided for under the TRIPS Agreement, may act as a barrier for making the results of scientific progress generally available.17 A UK government report suggested that the current system of intellectual property protections and patents has failed to stimulate research and development for neglected diseases, primarily as there is no profitable market for such drugs.18
There is consequently a greater need to invest public monies in the development of such drugs on public health, human rights, and moral grounds, or remove the need to recoup the costs of drug development through direct sales of that drug by providing other mechanisms for cost recovery. One such mechanism is the priority review voucher program which enables any pharmaceutical company that develops a new treatment for a neglected disease (including tuberculosis) to receive priority review from the US Food and Drug Administration for another product. A priority review voucher commits the FDA to reviewing a drug within six months, instead of the usual aim of ten months.19 While the authors of the original priority-review proposal suggest that a voucher could be worth more than 100 million USD, a recent sale of a voucher (which is transferrable) was recorded at 350 million USD.20 A priority review voucher was awarded to Janssen in 2012 for registering bedaquiline, potentially allowing the company to offset the costs of drug development for bedaquiline.21 This, and similar strategies have the potential to achieve delinkage and might be useful to explore in the future.
Of course, states can also leverage enhanced access to medicines through public-private partnerships, as has been effected by the Stop TB Partnership’s Global Drug Facility.22 However, these are initiatives limited to specific drugs on a case-by-case basis outside of any framework that provides strong obligations on any partners.
To date, there have been no cases before courts where the REBSP has been directly invoked aimed at forcing a government to make an available medicine accessible for the treatment of a particular disease. Similarly, there have been no cases where the REBSP has been used to leverage the development of new drugs for neglected diseases. This is due to its unknown nature, but also to the fact that it has not been recognized as a justiciable right that individuals can invoke directly. One rare example is a case from Argentina in which the right was not explicitly stated, but was implicitly at stake. A group of Argentinian NGOs brought the case, asking the state to manufacture a vaccine with known efficacy for the prevention of Argentine hemorrhagic fever, which affects people living in the high-risk pampas and who did not have easy access to preventive treatment. The private sector had failed to produce a drug, because it was not profitable to do so. The court ordered the state to manufacture the vaccine. The judgement was based, inter alia, on the right to health as included in the ICESCR.23 The number of international cases may increase in the future when the Optional Protocol to the ICESCR becomes more well-known as an international remedy for domestic victims of violations of the rights in the Covenant.24
From a more general perspective, therefore, it is apparent that the REBSP does not entitle individuals to direct enforceable benefits of scientific progress, but rather entails a right for people to have a legislative and policy framework adopted and implemented which aims at making the benefits of scientific progress available and accessible—both through encouraging new scientific discoveries, and through removing barriers for existing scientific knowledge to be used for public benefit. Steps to be taken in this regard must be deliberate, concrete, and as targeted as possible.25 This leads to the question of who has obligations or responsibilities to realize this right and what the nature of these obligations is, which we explore below using DR-TB to illustrate the general case.
MDR-TB burden and the status of drug treatment
Poverty is the sine qua non of TB. The majority of people in the world suffering from TB are poor and/or marginalized, and it disproportionately affects low-middle income countries with limited health care resources.26 Despite some progress in TB control, close to 10 million people are estimated to develop TB each year, of whom more than a third are not diagnosed and therefore not treated.27 The figures for MDR-TB are even more stark: only a quarter of estimated cases are detected, and fewer have access to treatment. While Eastern Europe has the world’s highest proportion of MDR-TB among all TB cases—up to 40% in some settings—the largest burden in terms of numbers lies in countries such as China, India, and South Africa, due to large populations and high TB incidence.28 While lack of effective tools for diagnosis of TB drug resistance remains a major barrier to effective MDR-TB treatment globally, there has been significant progress in this area.29
Most TB drugs in current use were developed between 1943 and 1963, and the current regimens involving combinations of first-line TB drugs have been in place for about 40 years. The reliance on the DOTS (Directly Observed Treatment, Short-Course) strategy, with extremely limited drug susceptibility testing and a limited armamentarium of available TB drugs, has left global TB programs exceptionally vulnerable to the rise of MDR-TB. Even with two new drugs recently approved for TB treatment (principally for MDR-TB treatment), the pipeline for new TB drugs remains poor.30 Prior to the approval of bedaquiline and delamanid, the most recent new TB drug to reach the market was rifampicin in the 1960s.31 Despite recent improvements, investment in TB drug research falls far short of what is estimated to be required, with the US$255 million in 2013 amounting to just a third of the annual target set in the Global Plan to Stop TB.32 Similarly, investment in global TB control falls short by approximately US$1.4 billion of that estimated to be required for effective implementation, including that required for MDR-TB treatment.33 Given the disproportionate burden of TB, and MDR-TB among the poor, the lack of investment in global strategies to combat TB and the consequent lack of a profitable market, there is a distinct lack of interest in TB drug development in the private sector. This results in two kinds of access problems: the lack of development of new drugs, on the one hand, and the inability to access new drugs (or newly repurposed drugs) for treating MDR-TB. Both represent challenges for thinking through any entitlement to benefit from scientific progress.
MDR-TB treatment currently requires at least four or five drugs, often with considerable side effects, for at least 18 months.34 Treatment includes an injectable agent, given daily, for between four and six months. Even under ideal conditions, treatment is successful for approximately 60% of patients; high mortality, treatment failure, and failure to complete the treatment regimen are common.35 It is therefore not surprising that the development of bedaquiline and delamanid, both new classes of drugs with TB activity, has raised hopes for a new, more efficacious, and more MDR-TB-tolerable treatment regimen.36 Despite regulatory approvals in a range of countries for both new drugs and available guidance on use from WHO, access to these drugs for MDR-TB patients has remained extremely limited.37 While bedaquiline received conditional approval from the US Food and Drug Administration (FDA) in 2012, and additional approvals in the European Union, South Africa, India, and other countries, less than 1,000 patients had received the drug by 2015.38 The situation for delamanid is even more dramatic; by February 2016, only 180 patients have been able to access the drug, primarily under compassionate use criteria.39 Delamanid was registered in Europe, Korea, and Japan in 2014, but no further registrations are pending, including in the countries contributing the majority of patients to the clinical trials on which drug registration was based.40
What is needed to improve MDR-TB treatment?
The reasons behind poor accessibility of these new drugs are multiple: high cost, potential side-effects leading to excess mortality, lack of systems for pharmacovigilance, resistance development, and, finally, lack of guidance on effective use of these new drugs for MDR-TB treatment in national TB programs. With bedaquiline priced at US$900 for a six-month treatment course, cost would appear to be a clear barrier, but even under a USAID-funded program to support bedaquiline access—where the drug was donated by the pharmaceutical company and is provided free to national TB programs—uptake has remained slow.41 Current guidance from WHO suggests that both bedaquiline and delamanid should be used as additional drugs in MDR-TB treatment, in cases where a treatment regimen cannot be constructed with existing drugs. In reality, this means that these new drugs are added to the existing, poorly tolerated, and lengthy treatment regimen, resulting in limited improvements to overall treatment success.42
Encouragingly, there are a small number of clinical trials underway and in planning that aim to incorporate bedaquiline, delamanid, or both into shorter, more efficacious MDR-TB treatment regimens.43 These regimens will require a combination of new, repurposed, and existing drugs in order to maximize efficacy. However, funding and capacity to conduct trials that are not directly linked to drug registration, that is, not funded by pharmaceutical companies, is limited. If the few trials currently underway or in planning produce promising results, these regimens then need to be made accessible in routine TB programs. While treatment regimens may be efficacious under clinical trial conditions, this may not translate into appropriate patient outcomes under programmatic conditions.44 Development and testing of new regimens—for example, of different duration and combinations in relation to likely adherence—would need to be implemented to ensure the medication is suitable for programmatic use, particularly in high-burden settings. As a result, ideally a continuous pipeline of new drug development should feed a constantly evolving clinical trial environment, the results of which are then operationalized in order to provide treatment for those in need. Thus, both access to existing drugs and the development of new drugs for MDR-TB are integrally related to each other, as well as representing key challenges for the REBSP.
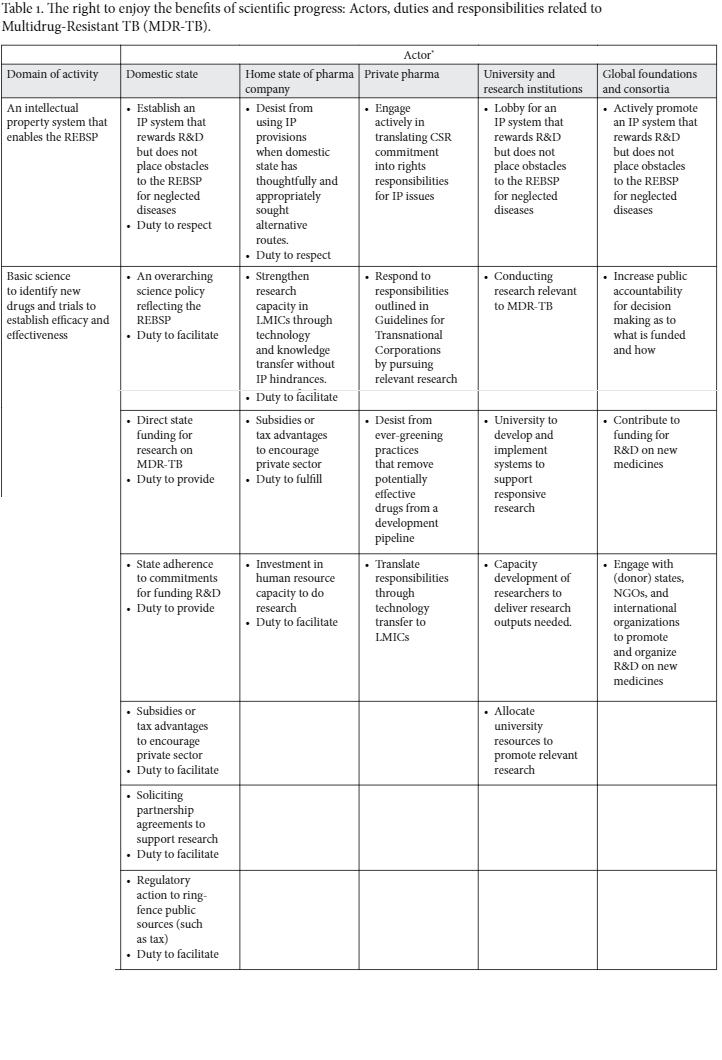
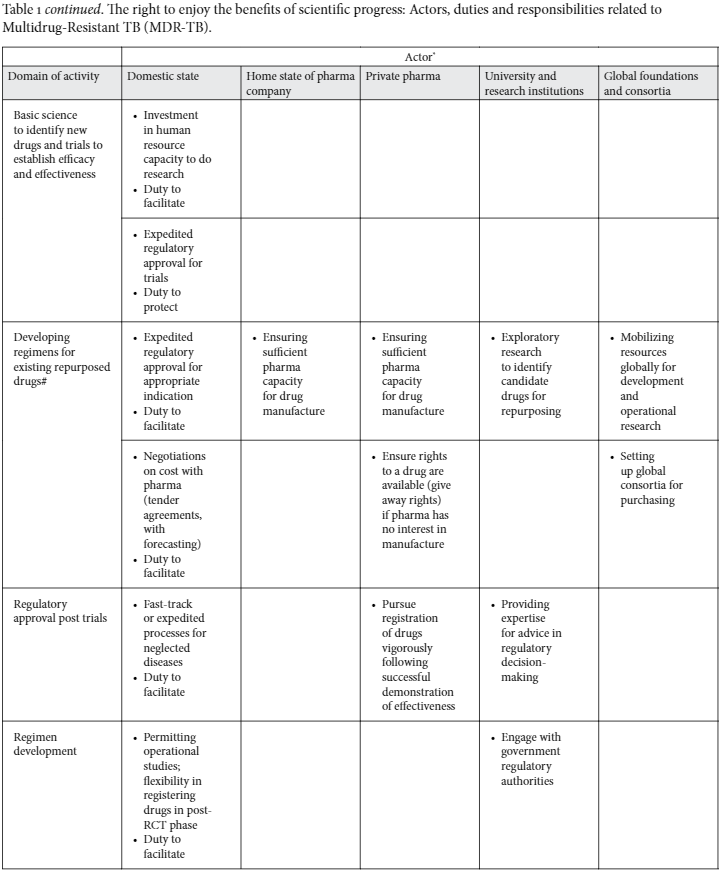
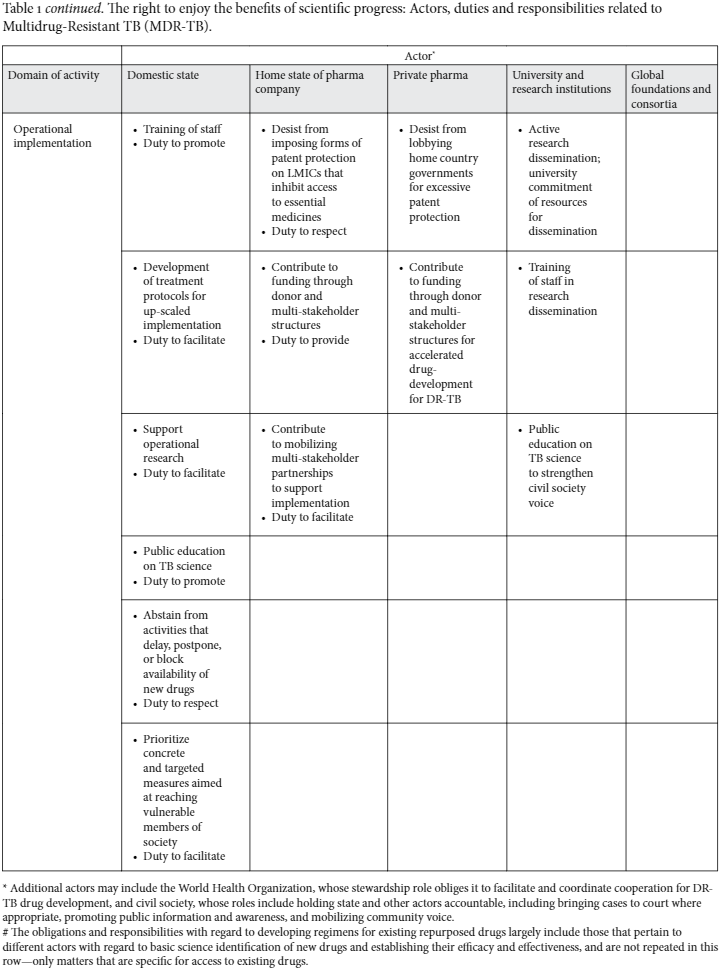
All along this pathway, important obstacles may be encountered, which, if translated into rights language, can generate particular obligations and responsibilities for different parties (Table 1).
The REBSP: obligations and responsibilities in relation to MDR-TB
When identifying the types of obligations that give substance to the REBSP, it makes sense to distinguish between different duty-holders and actors (Table 1). States who have voluntarily accepted to become legally bound by the ICESCR through ratification are the main duty-bearers. But is the state also the most appropriate actor to implement obligations resulting from Article 15.1b? In the 1960s, when the ICESCR was adopted, governments were centrally involved in funding research and the direct development of science, which was seen as a public good.45 This has changed drastically in present times, where the role and influence of the private sector (corporations and private research institutes) and non-state consortia has increased enormously and market forces often determine for which purposes both private and public resources will be spent.46 At the same time, the availability of public resources for research into new medicines has diminished.47
So what are the obligations of states today? The typology of human rights obligations (respect, protect, and fulfill) provides an appropriate framework for explanation.48 Obligations to respect the REBSP entail that both the domestic state and the home state of a pharmaceutical company must not introduce obstacles in its intellectual property laws and policy that would prevent institutes for research and development (R&D) from focusing on neglected diseases. The obligation to protect means that a state must regulate the conduct of corporations and prevent these from using their corporate power to frustrate the availability and accessibility of medicines that are not or less profitable. The human rights obligation to fulfill can be divided into obligations to facilitate and to promote. A state has an obligation to create an enabling environment for the development and implementation of a science policy which lays out principles that should underlie scientific research and establish an approach for research prioritization and diffusion of research findings.49 This is an example of an obligation to facilitate which is aimed at creating institutional arrangements to support the implementation of a right.50 With a view to making medicines for neglected disease available and accessible, states have an obligation to facilitate in the sense of stimulating research for new medicines by companies, research institutes, and universities, whether in the form of joint ventures or directly in the public sphere. This can be done, for example, by providing subsidies or tax advantages to incentivize research into neglected diseases, investing in the human resources base needed for research, and routing research grant funding into prioritized focus areas, whether through direct state funding of research, through partnership agreements with earmarked research funders, or through regulatory interventions to mobilize research funding. Direct funding for research is typically expressed in the form of a state commitment to allocate a minimum percentage of Gross Domestic Product (GDP) to R&D. For example, both the European Union and the Obama Administration have set spending targets for research and innovation at 3% of GDP.51 In 2006, The African Union adopted the Khartoum Declaration, which committed states to allocate a minimum of 1% of GDP to R&D. This was subsequently reinforced in the African Union’s Science, Technology and Innovation Strategy for Africa 2024.52 In practice, though, such commitments may be overly optimistic for low- and middle-income countries (LMIC). For example, of the 30 African countries that adopted the Abuja Declaration in 2001 to allocate 15% of total government expenditure to health, only one had reached this target 10 years later.53
The obligation to promote has a long-term nature: it aims at providing information and convincing key players of the need to make essential medicines available. It may be combined with public awareness-raising campaigns aimed at empowering the general public to make informed choices about their health, or at providing civil society and the public with information to pressure key stakeholders to act, as has been illustrated amply in the HIV treatment access arena.54
Additionally, once evidence of effectiveness is obtained, states should ensure that the regulatory approvals are expedited and systems for distribution put in place to put drugs into circulation as quickly as possible. For example, while South Africa’s Medicine Control Council approved a “clinical access” program for the new TB drug bedaquiline in January 2013, which removed delays in registering the drug, the cumbersome approval process for patients to access the drug still caused considerable delay in treatment initiation for extensively drug-resistant TB (XDR-TB).55 Even when regulatory approvals put drugs rapidly into the public domain, obstacles posed by patents that create unaffordable prices need addressing. MSF has shown how the patent held in South Africa for linezolid has prevented generic competition, kept the price high, and resulted in a failure to access linezolid for most patients in need, denying them the opportunity for possible cure.56 This challenges governments to pursue active purchasing programs able to negotiate prices down or, if needed, resort to the provisions of TRIPS that allow for parallel importation, generic substitution, or other exceptions to the exclusive rights of patent holders. For example, India’s Patent Act provides for the possibility to declare inventions not patentable with a view to prevent evergreening to ensure that patent protection is not granted for minor inventions.57 Moreover, a complex set of operational questions requires guidelines for how best to implement new regimens for MDR-TB that maximize access to new drugs of better efficacy, lower toxicity, and shorter duration, while avoiding development of further resistance through indiscriminate and poorly coordinated use of these medications.58 Guidelines to manage this competing pressure to open up access to new drugs while avoiding use patterns that increase the risk of new drugs developing resistance may require careful balancing of individual rights of access to care with the broader social good of protecting public health consistent with the Siracusa Principles.59 In South Africa, for example, state flexibility in allowing for off-schedule use of delamanid for compassionate use, and for operational research, has opened the possibility for salvage regimens to be operationally tested and to provide preliminary evidence for future programmatic guidelines, thus shortening the time to wider upscaling of treatment options in future. Thereafter, training of staff in new protocols should enjoy similar prioritization to ensure front-line staff is empowered to implement the new protocols carefully and avoid programmatic obstacles to implementation. Thus, states, as a part of their human rights obligations, should accelerate not only the research process but all the subsequent steps needed to see a new drug included in standard treatment protocols. It is noted that patents regimes may frustrate or delay this endeavor in many countries.60
From a normative perspective, therefore, the REBSP entails a right for individuals to have a legislative and policy framework adopted that enables the development and manufacturing of a new medicine. This means both that the policy should not impede or interfere with such drug development and/or repurposing for DR-TB treatment (respect), but also that it should ensure corporations do not delay new knowledge for medicines for neglected diseases from becoming available (protect) and put in place positive measures to encourage drug development and/or repurposing (fulfill).61Such a policy framework must be reasonable in conception and implementation, which means that it must be coordinated and coherent with a view to focusing on the need to make new and repurposed medicines available for diseases that disproportionately affect the poor, who would otherwise be denied access. For patients with DR-TB, therefore, such a framework is informed by a human rights approach by which the state would be expected to prioritize measures in its science policy that give preference to drug development and/or repurposing for neglected diseases that disproportionately affect poor and vulnerable members of society.
Rights claims in a globalizing society
In an era characterized by economic globalization, however, it is self-evident that making essential medicines available is a process involving multiple actors: not only pharmaceutical companies, but also their home states, international organizations, and private funders (Table 1). Lack of availability of essential medicines for the treatment of neglected diseases is a global problem that demands a global response. It has been argued that especially governments in the Global North who have the capacity and resources have an obligation to contribute to the fulfillment of human rights outside of their national boundaries.62 Through processes of economic globalization, actions and omissions by actors in one state may affect the human rights of people in other countries. However, traditionally, human rights law only applied on the territory of states that have ratified human rights treaties. With a view to filling this normative gap, there is debate over the establishment of a human rights framework that applies beyond national borders. Such a framework would contain both negative and positive obligations. This is the idea of extraterritorial human rights obligations aimed at realizing the universal scope of human rights, a concept elaborated in the Maastricht Principles on the Extraterritorial Obligations of States in the Area of Economic, Social and Cultural Rights. Drawn from international law, these principles also aim to clarify the content of extraterritorial state obligations for contributing to the fulfillment of the right to health in countries with serious public health problems.63
An extraterritorial human rights obligation for states in the Global North resulting from the REBSP would logically take two forms. First, states should desist from policies that undermine the rights of LMIC populations to enjoy the benefits of scientific progress (respect). One example is overzealous insistence on patent protection and pursuance of pharmaceutical company pricing protections in recipient countries at the expense of local access to medicines; this was demonstrated in developed countries’ behavior in the WTO in the lead-up to the Doha Declaration. More recently, it is also evident in the conclusion of numerous bi- and multilateral (TRIPS-plus) trade agreements subsequent to Doha which continue to undermine DOHA TRIPS flexibilities and the ability of poorer countries to develop their own IP regimes.64 Patent protection has been shown to be associated with inappropriately high costs of medications which then inhibit governments from adopting efficacious but costly medicines in their regimens.65 Notably, paragraph 3.b of the Sustainable Development Goals recognizes the need to support R&D for vaccines and medicines that primarily affect developing countries and to provide access to such medicines under the Doha Declaration.66
Second, developed countries might well be expected to realize this obligation by strengthening the research infrastructure and capacity in the Global South through promoting the transfer of scientific knowledge free from intellectual property rights.67 A division and coordination of domestic and extraterritorial human rights obligations between states is required, depending on the level of development, the domestic health situation and TB prevalence, and the capacity and resources of more wealthy states to assist.68
Third, another potential avenue is that represented by funds set up for drug and vaccine development as global collaborations with contributions from rich countries, the corporate sector, and donor organizations.69 In this regard, there is a need to mobilize and share common but differentiated responsibilities of all relevant stakeholders, both governmental and non-governmental, for international cooperation aimed at giving effect to the right to essential medicines.70 However, thus far, states from the North appear unwilling to accept that they also have human rights obligations beyond their national borders on the basis that such a commitment would be contrary to their interests and would limit their policy freedom abroad.71 Ironically, these same states emphasize the importance of LMICs complying with intellectual property standards so as to protect the interests of their pharmaceutical industries abroad. The bilateral and multilateral TRIPS-plus agreements are a good example of this trend.72
Nonetheless, pharmaceutical companies are key players for making essential medicines available. They have the human and financial resources and R&D capacity. Although corporations do not have human rights obligations, they do have special functions and responsibilities to engage in activities for the promotion of public health. In this regard, corporate social responsibility can be made concrete by giving substance to the implementation of the REBSP. As Lee and Hunt have argued, pharmaceutical companies have a shared right to health responsibility to engage in R&D for neglected diseases that affect poor people in poor countries.73 It is what society may expect from having “a social license to operate” for the general welfare, not just for profit. People who suffer from MDR-TB have only one option for accessing drugs, so their treatment success is partly reliant on the actions of these companies. These companies may therefore be said to perform a public function that the state is unable to execute.
There is also a body of work developing that outlines the responsibilities of corporations for meeting human rights standards. Initially framed as norms for transnational corporations equivalent to rights obligations of governments, but subsequently reframed as guiding principles after considerable resistance to the accountability associated with the norms approach, these principles recognize “the role of business enterprises as specialized organs of society performing specialized functions, required to comply with all applicable laws and to respect human rights.”74 Both arguments point to the need for pharmaceutical companies to operate within a human rights framework, albeit with lesser levels of accountability than governments.
While efforts have been directed to identifying the role of corporations in a rights-based approach to health, there are many other non-state actors involved. Universities and research institutions, typically funded with public moneys, but with relative autonomy from the state, are instrumental in conducting both the basic and the applied research needed to bring new drugs into the development pipeline, and to identify drugs with repurpose uses. In the same way that for-profit corporations may have legitimate expectations placed upon them as a social license to operate, universities are increasingly being called on to demonstrate their social responsiveness.75Here one might find a trade-off in balancing academic freedom for the researcher to pursue whatever line of inquiry they find academically compelling (academic freedom itself being a right protected in Article 15.3 of the ICESCR) against the expectation that universities focus their research in ways that prioritize the most urgent societal challenges. Clearly, researchers cannot be compelled to engage in research to develop new TB drugs, but, in practice, generous funding streams and academic reward systems responding to priority areas such as MDR-TB could and do influence researcher behavior and direct researcher focus. For example, the influence of donor funding in prioritizing HIV and TB-related research has been shown in the pattern of research conducted in many settings across the world.76
However, it is typically in the translation of scientific research where universities lag, and where the REBSP highlights important gaps. Ensuring that scientific findings reach audiences who could act to ensure the implementation of the findings is often a responsibility omitted from university reward systems, and often unfunded by donors. Such audiences are typically policy-making structures both in the executive and legislature, but should also include relevant civil society actors, whose agency may be key to realizing rights and to ensuring the policies and programs for implementation of new scientific knowledge.77
The experience in the HIV field is instructive, where very strong civil society action spurred research and development of new drugs, raised awareness of entitlements to treatment among those most affected by the HIV pandemic, and directly impacted on drug pricing for ARVs to treat HIV.78 The close alliance of scientists with the treatment access movement in South Africa played a key role in a number of important public health advances and in advancing access to the benefits of scientific research related to HIV.79 Thus, the obligation on researchers to engage actively in the dissemination of their findings and in the conveying of scientific information to non-scientific audiences represents not only good scientific practice, but a strengthening of the roles of universities and research institutions to help realise the REBSP. If taken seriously, universities would then need to invest in training researchers in better public engagement in science, rewarding scientific activities that enhance popular understanding of science, and dissemination activities aimed at ensuring research findings are translated into practical change.
Lastly, global health governance has undergone massive changes in the past two decades. The WHO was previously seen as the undisputed steward of matters affecting health on a global scale; this has changed radically in the past 20 years, with the growth of large private and public-private consortia and agencies whose funding mandates are so large that they dwarf that of WHO. Questions have been raised about the accountability of these foundations and to what extent they have turned global health governance into a terrain dominated by rich and powerful interests.80 In the same way private corporations are being asked hard questions regarding accountability and standards, we believe the same principles should apply to these newer agencies of global health governance. They have a key role in realizing the right to health and in ensuring people’s REBSP, which, rather than being framed as philanthropy or largesse, should be seen within the unfolding context of the extended purview of rights frameworks beyond national boundaries and to non-state actors. Civil society organizations (CSOs) and patient groups should persuade corporations to take up their social responsibility in this area.
To some extent, we have seen that collaboration between the corporate sector and university research institutes may help to bridge the gap between basic science and clinical research to advance innovative medicines for poor patients, for which we already have some multi-stakeholder models such as the Drugs for Neglected Diseases Initiative and Critical Path to TB Drug Regimens.81 Financing mechanisms, such as illustrated in the TB Alliance, are also putting teeth to multi-stakeholder initiatives to accelerate drug development, which will enhance availability and accessibility consistent with the REBSP. Nonetheless, questions about the implications of locating such hugely important policy initiatives outside of a framework of state sovereignty (and the associated system of human rights-based accountability) remain potential threats to a full realization of the REBSP.82
Conclusion
It is clear that the REBSP does not provide an individual entitlement to claim a right to have a new drug developed for a neglected disease such as MDR-TB. However, it does provide a number of important levers complementary to right to health claims. It sets up entitlements for a system of science development and dissemination that could and should benefit the most marginalized in society, and represents a collective claim for the use of science for social benefits. Inasmuch as the market fails to address neglected diseases such as MDR-TB, the REBSP provides a potential counterbalance to frame a positive obligation on states to both marshal their own resources and to coordinate the actions of multiple other actors towards this goal. Spanning the full gamut, these are obligations of respect (to refrain from policies that violate rights), protect (regulate the behavior of private actors) and fulfill (generate an enabling policy environment for priority research). At the same time, non-state actors can be brought within the ambit of contributing to addressing one of the major public heath crises of the current period without holding the same level of accountability as states but still recognizing obligations at a level of ‘soft law’ responsibilities.
We believe that there are enough grounds to warrant testing the REBSP framework further in different case settings—both in relation to neglected diseases and other access-to-health contexts. For example, new technologies carry the promise of screening for a range of non-communicable diseases or for new forms of safer production that reduce risks for workers and consumers from hazardous chemicals. Developing the conceptualization of the REBSP and advancing the evidence base for its role may provide powerful practical tools for human rights advocates, civil society activists, and health and human rights researchers to tackle neglected diseases and address wider health challenges across the globe.
Acknowledgments
Funding for this work was obtained from the University of Cape Town in terms of a University of Cape Town International Collaborative Research Grant.
Leslie London, MD, is Professor of Public Health Medicine and head of the Health and Human Rights Program in the School of Public Health and Family Medicine, University of Cape Town, South Africa.
Helen Cox, PhD, is a Wellcome Trust Intermediate Fellow in the Division of Medical Microbiology, Department of Pathology, University of Cape Town, South Africa.
Fons Coomans, PhD, is Professor of Human Rights at the Faculty of Law of Maastricht University, The Netherlands, and a Visiting Professor at the School of Public Health and Family Medicine, University of Cape Town, South Africa.
Please address correspondence to the authors c/o Leslie London, School of Public Health and Family Medicine, University of Cape Town Health Sciences Faculty, Anzio Rd, Observatory, 7925, Cape Town, South Africa. Email: leslie.london@uct.ac.za
Competing Interests: None declared.
Copyright: London, Cox, and Coomans. This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/), which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
References
- Chapman, “Towards an Understanding of the Right to Enjoy the Benefits of Scientific Progress and its Applications,” Journal of Human Rights 8/1 (2009), pp. 1-36; Y. Donders, “The right to enjoy the benefits of scientific progress: in search of state obligations in relation to health”, Medicine Health Care and Philosophy 14 (2011), pp. 371-381; Farida Shaheed, UN Special Rapporteur in the field of cultural rights, The Right to Enjoy the Benefits of Scientific Progress and Its Applications, UN Doc. No. A/HRC/20/26 (2012).
- World Health Organization. 2015. Global tuberculosis report 2015. World Health Organization: Geneva; S. Keshavjee and P. E. Farmer, “Time to put boots on the ground: making universal access to MDR-TB treatment a reality, ”The International Journal of Tuberculosis and Lung Disease 14/10 (2010), pp. 1222-1225; S. Keshavjee and P. E. Farmer, “Picking up the pace–scale-up of MDR tuberculosis treatment programs,” New England Journal of Medicine 363 (2010), pp. 1781-1784; C. Y. Chiang, R. Centis and G. B. Migliori, “Drug-resistant tuberculosis: past, present, future,” Respirology 15/3 (2010), pp. 413-432; N. R. Gandhi, P. Nunn, K. Dheda, et al., “Multidrug-resistant and extensively drug-resistant tuberculosis: a threat to global control of tuberculosis,” The Lancet 375/9728 (2010), pp. 1830-1843.; A. Matteelli, R. Centis, L. D’Ambrosio and G. B. Migliori, “Multidrug-resistant tuberculosis today,” Bulletin of the World Health Organization 90/2 (2012), p. 78.
- World Health Organization (see note 2).; I. Abubakar, M. Zignol, D. Falzon, et al., “Drug-resistant tuberculosis: time for visionary political leadership,” The Lancet Infectious Diseases 13/6 (2013), pp. 529-539; E. A. Kendall, M. O.Fofana, D. W. Dowdy, “Burden of transmitted multidrug resistance in epidemics of tuberculosis: a transmission modelling analysis,” Lancet Respiratory Medicine 3/12 (2015), pp. 963-972.
- World Health Organization ( see note 2).
- G. Brigden, B. T. Nyang’wa, P. du Cros, et al., “Principles for designing future regimens for multidrug-resistant tuberculosis,” Bulletin of the World Health Organization 92/1 (2014), pp. 68-74.
- J.C. Johnston, N.C. Shahidi, M. Sadatsafavi et al., “Treatment outcomes of multidrug-resistant tuberculosis: a systematic review and meta-analysis,” PLoS ONE 4/9 (2009), p. e6914.
- Keshavjee et al. ( see note 2).
- J.M. Burgers, “Rights and Duties Concerning Creative Expression in Particular in Science,” in: UNESCO, Human Rights: Comments and Interpretations, UNESCO/PHS/3 (rev.), (1948), p. 216.
- Chapman, (see note 1), pp. 1-36. 10. Ibid., p. 9; Shaheed (see note 1), para. 26.
- UNESCO Recommendation on the Status of Scientific Researchers (1974), para. 1a. Available at http://www.unesco.org.
- Committee on Economic, Social and Cultural Rights, General Comment No. 3, the Nature of States Parties Obligations, UN Doc. No. E/1991/23, Annex III, para. 9.
- L. London, C. Himonga, N. Fick et al., “Social solidarity and the right to health: essential elements for people-centred health systems,” Health Policy and Planning 30 (2015), pp. 938-945.
- Committee on Economic, Social and Cultural Rights, General Comment No.14, the Right to the highest Attainable Standard of Health, UN Doc. E/C.12/2000/4 (2000), para. 59. Available at http://tbinternet.ohchr.org/_layouts/treatybodyexternal/TBSearch.aspx?Lang=en&TreatyID=9&DocTypeID=11.
- Ibid., para. 12.
- See J. Sellin, Access to Medicines – The interface between patents and human rights (Antwerp/Oxford: Intersentia, 2014), chapter 5.
- World Trade Organisation. Agreement on Trade-Related Aspects of Intellectual Property Rights, Part II, section 7 on Protection of Undisclosed Information. Available at https://www.wto.org/english/tratop_e/trips_e/t_agm3_e.htm#7.
- Commission on Intellectual Property Rights, Integrating Intellectual Property Rights and Development Policy (London 2002). Available at: http://www.iprcommission.org/graphic/documents/final_report.htm.
- J. Moe, H. Grabowski, and D. Ridley, FDA review vouchers. N Engl J Med, 2009. 360 (8): p. 837; author reply 837-8.
- United Therapeutics, United Therapeutics Corporation Agrees to Sell Priority Review Voucher to AbbVie for $350 Million (United Therapeutics, August 2015). Available at http://ir.unither.com/releasedetail.cfm?ReleaseID=928100
- FDA, CDER, CBER (compiled by), Guidance for Industry Expedited Programs for Serious Conditions – Drugs and Biologics (May 2014). Available at www.fda.gov/downloads/drugs/…/guidances/ucm358301.pdf.
- Stop TB Partnership, Stop TB Partnership’s Global Drug Facility jumpstarts access to new drugs for MDR-TB with innovative public-private partnerships (February 2016). Available at http://us3.campaign-archive1.com/?u=85207b84f0f2d8ddc9bd878de&id=a2d0bb121e.
- C. Mariela and V. Viceconte. Ministry of Health and Social Welfare (1998) Case no. 31.777/96, June 2, 1998.
- Optional Protocol to the International Covenant on Economic, Social and Cultural Rights, Doc. No. A/63/435 C.N.869.2009.TREATIES-3 (2008). Available at http://www.ohchr.org/EN/ProfessionalInterest/Pages/OPCESCR.aspx.
- Committee on Economic, Social and Cultural Rights, General Comment No. 3, The Nature of States Parties’ Obligations, UN Doc. E/1991/23, Annex III, para. 2.
- O. Oxlade, M. Murray, “Tuberculosis and poverty: why are the poor at greater risk in India?” PLoS One 7/11 (2012), p. e47533.
- World Health Organization (2015, see note 2).
- Ibid., 2015.
- S. E. Dorman, “New Diagnostic Tests for Tuberculosis: Bench, Bedside, and Beyond,” Clinical Infectious Diseases 50/S3 (2010), pp. S173-S177.
- P. Clayden, S. Collins, M. Frick, et al., Drugs Diagnostics, Vaccines, Preventive Technologies, Research toward a cure, and Immune-based and Gene Therapies in Development (Treatment Action Group Pipeline Report, July 2015). Available at www.treatmentactiongroup.org/pipeline-report.
- Z. Ma, C. Lienhardt, H. McIlleron et al., “Global tuberculosis drug development pipeline: the need and the reality,” Lancet 375/9731 (2010), pp. 2100-2109.
- Clayden (see note 30).
- World Health Organization (2015, see note 2).
- Ibid, 2015; S. Wu, Y. Zhang, F. Sun et al., “Adverse Events Associated with the Treatment of Multidrug-Resistant Tuberculosis: A Systematic Review and Meta-analysis,” American Journal of Medicine 23/2 (2013), p. e521-530.
- S. D. Ahuja, D. Ashkin, M. Avendano et al., “Multidrug resistant pulmonary tuberculosis treatment regimens and patient outcomes: an individual patient data meta-analysis of 9,153 patients,” PLoS Medicine 9/8 (2012), p. e1001300;
- Brigden (see note 5).
- World Health Organization, The use of Bedaquiline in the treatment of multidrug-resistant tuberculosis (WHO, 2013). Available at www.who.int/tb/challenges/mdr/bedaquiline/en/; World Health Organization, WHO Interim Guidance on the use of Delamanid in the treatment of MDR-TB (Barcelona: WHO, 2014). Available at www.who.int/tb/features_archive/delamanid/en/.
- Clayden (see note 30); Médecins Sans Frontières, Ready, set, slow down: new and promising TB drugs are grabbing headlines but not reaching patients (MSF, March 2015).
- Médecins Sans Frontières, Developing countries hit with high price for important new tuberculosis drug (MSF, Feb 2016).
- Clayden (see note 30).
- USAID, USAID’s Bedaquiline Donation Program in Partnership with Johnson and Johnson (USAID, Feb 2016). Available at https://www.usaid.gov/what-we-do/global-health/tuberculosis/technical-areas/bedaquiline-donation-program.
- H. S. Cox, J. J. Furin, C. D. Mitnick et al., “The need to accelerate access to new drugs for multidrug-resistant tuberculosis,” Bulletin World Health Organization 93/7 (2015), pp. 491-497.
- Resist TB, DR-TB Clinical Trials Progress Report (Resist TB, Feb 2016). Available at http://www.resisttb.org/?page_id=1602.
- Cox et al. (2015, see note 42).
- Chapman (see note 1), p. 8.
- D. Sanders, F. E. Baum, A. Benos, et al., “Revitalising primary healthcare requires an equitable global economic system – now more than ever,” Journal of Epidemiology and Community Health 65/8 (2011), pp. 661-665; A. R. Katz, “Noncommunicable diseases: global health priority or market opportunity? An illustration of the World Health Organization at its worst and at its best,” International Journal of Health Services 43/3 (2013), pp. 437-458; D. Callahan and A. A. Wasunna, Medicine and the Market: Equity v. Choice, (Baltimore, Maryland: The Johns Hopkins University Press, 2006), pp 117-202.
- J. Chakma, G. H. Sun, J. D. Steinberg et al., “Asia’s Ascent — Global Trends in Biomedical R&D Expenditures,” New England Journal of Medicine 370/1 (2014), pp. 3-6; E. Dorsey, J. de Roulet, J. P. Thompson, et al. “Funding of US Biomedical Research, 2003-2008,” Journal of American Medical Association 303/2 (2010), pp.137-143. doi:10.1001/jama.2009.1987
- M. Sepúlveda, The Nature of the Obligations under the International Covenant on Economic, Social and Cultural Rights, (Antwerp/Oxford: Intersentia, 2003), chapter V.
- Shaheed, (see note 1), p. 13.
- Sepúlveda, (see note 48), pp 239-241.
- European Commission, The European Union explained: Research and Innovation (European Union: Brussels, 2014). Available at http://europa.eu/pol/rd/index_en.htm; National Economic Council, Council of Economic Advisers, and Office of Science and Technology Policy, A Strategy for American Innovation. Securing Our Economic Growth and Prosperity (The White House: Washington DC, 2011). Available at https://www.whitehouse.gov/sites/default/files/uploads/InnovationStrategy.pdf.
- African Union, Khartoum Decision EX.CL/Dec.254 (VIII) (2006). Available at http://www.au.int/en/sites/default/files/EX%20CL%20DEC%20236%20-%20277%20%28VIII%29%20_E.pdf; African Union, Science, Technology and Innovation Strategy for Africa 2024 (2014). Available at http://hrst.au.int/en/sites/default/files/STISA-Published%20Book.pdf.
- World Health Organization, The Abuja Declaration: Ten Years On (Geneva: WHO, 2011). Available at http://www.who.int/healthsystems/publications/abuja_report_aug_2011.pdf.
- Sepúlveda (see note 50), pp 243-246; General Comment 14 (see note 14), para. 37; M. Heywood, “South Africa’s Treatment Action Campaign: Combining Law and Social Mobilization to Realize the Right to Health,” Journal of Human Rights Practice 1/1 (2009), pp. 14-36.
- Médecins Sans Frontières,, DR-TB drugs under the microscope. Sources and prices for Drug-Resistant Tuberculosis medicines (MSF: Brussels, 3rd Edition – October 2013).
- Médecins Sans Frontières, Linezolid Fact Sheet (MSF, June 2014).
- See for a discussion of India’s Patent Act (section 3d) and the Novartis Case, Sellin (see note 16), pp.369-377.
- Médecins Sans Frontières, MSF Crisis Alert: The new face of an old disease: urgent action needed to tackle global drug-resistant TB threat (MSF: Brussels, 2014); A. Matteelli, A. Roggi and A. C. Carvalho, “Extensively drug-resistant tuberculosis: epidemiology and management,” Clinical Epidemiology 1/6 (2014), pp. 111-118.
- L. London, “Confinement for extreme drug-resistant TB (XDR-TB): Balancing protection of health systems, individual rights and the public’s health,” International Journal of Tuberculosis and Lung Disease 13/10 (2009), pp. 1200-1209.
- Médecins Sans Frontières, Spotlight on Trans Pacific Partnership Agreement (MSF, January 2016). Available at http://www.msfaccess.org/spotlight-on/trans-pacific-partnership-agreement.
- Shaheed, (see note 1).
- General Comment No. 14 (see note 14), para. 39; International Council on Human Rights Policy, Duties sans Frontières – Human rights and global social justice (Geneva: International Council on Human Rights Policy, 2003).
- Maastricht Principles on Extraterritorial Obligations of States in the Area of Economic, Social and Cultural Rights (September 2011). See in particular the Preamble and Principles 8 and 28-32. Available at http://www.etoconsortium.org.
- Sellin (see note 16), pp. 281-288.
- World Health Organization, The World Medicines Situation 2011. Medicines Prices, Availability and Affordability (Geneva: WHO, 2011).
- Sustainable Development Knowledge Platform, Transforming our World: the 2030 Agenda for Sustainable Development (2015). Available at https://sustainabledevelopment.un.org/post2015/transformingourworld.
- Shaheed, (see note 1), para. 68.
- Maastricht Principles (see note 62), Principle 30, 31; See also M.E. Salomon, Global Responsibility for Human Rights (Oxford: Oxford University Press, 2007), chapter 5
- See M. Balasegaram, C. Bréchot, J. Farrar, et al., “A Global Biomedical R&D Fund and Mechanism for Innovations of Public Health Importance”. PLoS Medicine 12/5 (2015), p. e1001831.
- M. Salomon, Global Responsibility for Human Rights (Oxford: Oxford University Press, 2007), p. 193; See for example the Stop TB Partnership, http://www.stoptb.org.
- M. Langford, F. Coomans, and F. Gómez Isa, “Extraterritorial Duties in International Law”, in: M. Langford, W. Vandenhole, M. Scheinin, W. van Genugten (eds.), Global Justice, State Duties (Cambridge: Cambridge University Press, 2013), pp. 62-65.
- Sellin (see note 16).
- J.Y. Lee, P. Hunt, “Human Rights Responsibilities of Pharmaceutical Companies in Relation to Access to Medicines,” Journal of Law, Medicine & Ethics (2012), pp. 220-233; See also Human Rights Guidelines for Pharmaceutical Companies in relation to Access to Medicines, drafted by Paul Hunt, UN Special Rapporteur on the right of everyone to the enjoyment of the highest attainable standard of physical and mental health, UN Doc. No. A/63/263, Annex (2008).
- United Nations, Guiding Principles on Business and Human Rights: Implementing the United Nations “Protect, Respect and Remedy” Framework, UN Doc. No. A/HRC/17/31. Available at http://www.ohchr.org/Documents/Issues/Business/A-HRC-17-31_AEV.pdf.
- “Talloires Declaration on the Civic Roles and Social Responsibilities of Higher Education,” (Presentation at Talloires Conference, September 2005). Available at http://www.vosesa.org.za/focus/vol1_no2/Talloires_Declaration_2005.pdf.
- L. London, T. Naledi, S. Petros, “Health research in the Western Cape Province, South Africa: Lessons and challenges,” African Journal of Primary Health Care and Family Medicine 6/1 (2014), Art. #698, 7 pages. Available at: http://www.phcfm.org/index.php/phcfm/article/view/698; L. Shiffman.”Has donor prioritization of HIV/AIDS displaced aid for other health issues?” Health Policy and Planning, 23/2 (2008), p.p. 95-100; G. Lordan, K. K. Tang, F. Carmignani. “Has HIV/AIDS displaced other health funding priorities? Evidence from a new dataset of development aid for health,” Social Science and Medicine, 73/), (2011), p.p. 351-5.
- L. London, “Issues of equity are also issues of rights: Lessons from experiences in Southern Africa,” BioMed Central Public Health 7 (2007), p.14. Available at: http://www.biomedcentral.com/content/pdf/1471-2458-7-14.pdf.
- Clayden (see note 30); Heywood (see note 54).
- M. Heywood, “South Africa’s Treatment Action Campaign: Combining Law and Social Mobilization to Realize the Right to Health,” Journal of Human Rights Practice 1/1 (2009), pp. 14-36; N. Geffen, Debunking Delusions: The inside story of the TAC. Auckland Park: Jacana Media, 2010).
- L. Haynes, D. Legge, L. London, et al., “Will the struggle for health equity and social justice be best served by a Framework Convention on Global Health?” Health and Human Rights 15/1 (2013), pp. 111-116. Available at http://www.hsph.harvard.edu/hhrjournal/wp-content/uploads/sites/2469/2013/06/Haynes-FINAL.pdf; I. Kickbusch and M. M. Szabo, “A new governance space for health,” Global Health Action 7 (February 2014), p. 23507.
- University of Cape Town, Collaboration with pharmaceutical giant to foster drug development in Africa (UCT Monday Paper, March 2013). Available at http://www.uct.ac.za/mondaypaper/archives/?id=9498; and see Drugs for Neglected Diseases Initiative, http://www.dndi.org; see Critical Path to TB Drug Regimes, http://www.cptrinitiative.org/.
- Haynes et al., (see note 80).