Falling Short of the Rights to Health and Scientific Progress: Inadequate TB Drug Research and Access
Mike Frick, Ian Henry, and Erica Lessem
Abstract
The incorporation of human rights-based approaches into TB programs is gaining traction, but little work has explored the application of human rights norms and principles to TB research (a domain traditionally left to bioethics). TB research is gravely underfunded, and the scarcity of resources for TB drug development has contributed to the stubborn persistence of the TB epidemic and helped to create the conditions under which drug-resistant TB has developed and spread. This article shows how human rights—particularly human rights standards, norms, and principles related to the rights to health and benefits of scientific progress—can provide insight into understanding how underfunding TB drug research undermines efforts to secure access to safe, effective, and optimized treatment for all people with TB. By analyzing TB research in relation to the rights to health and scientific progress, we aim to clarify the legal obligations of governments to improve the TB drug research system, fund TB research, and make medical advances that result from research available to all people with TB.
Introduction
Tuberculosis (TB) has been preventable, treatable, and curable with combination drug therapy since the 1950s, but more than six decades later continues to claim 1.5 million lives a year.1 The limitations of current treatment options, and the slow pace of efforts to develop newer, better ways of treating TB, are leading factors driving the global TB epidemic, particularly in the face of rising rates of drug resistance—a human-made phenomenon. In this paper, we first discuss three treatment-related inadequacies that have hindered the response to TB: 1) inadequate options to treat TB; 2) inadequate financing for research and development (R&D) to develop better options; and 3) inadequate access to treatment options, both new and old. We argue that these shortcomings in research and access, commonly attributed to weak market incentives and the absence of political will, are the result of states’ failures to uphold their obligations under international human rights law. In particular, we examine states’ obligation to support TB research as part of meeting their commitments to fulfill the right to the highest attainable standard of health (for example, Article 12 of the International Covenant on Economic, Social and Cultural Rights or ICESCR) and the right to enjoy the benefits of scientific process and its applications (for example, Article 15 of the ICESCR).2
Considered together, the rights to health and scientific progress provide a framework—grounded in state-level legal obligations—for linking the financing, conduct, and ownership of TB research to the challenges many people with TB face in accessing adequate treatment. Under international human rights law, access is composed of multiple considerations, and “the dimensions of access require some adaptability from right to right.”3 For the right to health, General Comment 14 of the Committee on Economic, Social, and Cultural Rights (CESCR) identifies four overlapping components of access: non-discrimination, physical accessibility, economic accessibility (affordability), and access to information.4 These dimensions of accessibility join availability, acceptability, and quality of health facilities, goods, and services as essential elements of the right to health in all its forms and at all levels.5
Access considerations also form the “cornerstone” of the right to science.6 The earliest formulation of this right in Article 27 of the Universal Declaration of Human Rights points toward the right of all people to take part in cultural life, enjoy the arts, and share in the benefits of scientific progress.7 The drafting history of ICESCR Article 15 makes clear that access in this context entails enjoying the actual—and in the case of health technologies like medicines, tangible—applications of scientific progress (that is, access to the benefits of scientific progress go beyond mere sharing in the diffuse benefits that accrue from general scientific advancement).8 The text of Article 15 establishes the obligation of states to take steps “necessary for the development and diffusion of science and culture.”9 By speaking of development and diffusion together, Article 15 offers a concept of access that connects state support for research and innovation (development) to the obligation of states to ensure that all people enjoy the benefits of science and its applications without discrimination (diffusion).10 The inadequacies of TB treatment illustrate how fulfilling the rights to health and scientific progress require states to support TB research and structure this support in ways that promote both the development and diffusion of new and improved TB drugs.
Inadequate treatment options for TB
The majority of drugs used to treat TB are decades old, and several have never been studied for TB under the rigorous conditions of randomized, controlled trials. Only two new drugs from new drug classes have been approved to treat TB in the last 40 years. Even with these two advances, treatment for all forms of TB is long and comes with high pill burdens. Patients with drug-susceptible TB (DS-TB) take treatment for six months, while those with drug-resistant TB (DR-TB) must endure treatment for up to two years. During this time, the typical multidrug-resistant TB (MDR-TB) patient will receive 240 painful drug injections and swallow 14,600 pills.11 Many of the drugs used to treat MDR-TB are poorly tolerated and carry the potential for serious—and in some cases irreversible—toxicities, including peripheral neuropathy, hearing loss, and psychosis. Even then, the effectiveness of MDR-TB treatment is low, with global cure rates persisting at 48%.12 Poor MDR-TB cure rates are attributable not only to the challenges of completing therapy (such as pill burden, toxicities, and length of treatment), but also to the poor anti-TB activity of the drugs themselves. The majority of drugs the World Health Organization (WHO) recommends to treat MDR-TB are not even demonstrated to be bactericidal (capable of killing bacteria), but rather are added onto a backbone of other drugs to protect against the amplification of resistance.13
Over time, the limitations of TB treatment have changed the nature of TB disease itself, whereby drug-sensitive strains have become chronic and deadly by developing resistance to existing antibiotics. By leaving patients reliant on lengthy, difficult-to-tolerate regimens that complicate adherence, the weak R&D environment has helped to create the conditions under which DR-TB strains arise and are transmitted in communities.14 In other words, the failures of the R&D system have contributed to the successful adaptation of the TB pathogen, and have continued to permit its flourishing, at the expense of human health and life. This biosocial phenomenon has been driven by human activity, but the attribution of responsibility has wrongly zeroed in on TB patients themselves.15 The oft-voiced idea that most drug resistance results when patients fail to adhere to therapy is challenged by research tracing a majority of DR-TB cases in some settings to primary transmission.16 In addition, whole-genome sequencing of clinical isolates from DR-TB outbreaks suggest that “drug-resistant strains circulating today reflect not only vulnerabilities of current TB control efforts but also those that date back 50 years.”17 This evidence underscores the need to shift the frame of analysis away from the behaviors of individual TB patients today to focus on the continuing legacy of government failures to prevent and respond to DR-TB by providing adequate treatment. By focusing on the obligations of governments, human rights offer a more suitable framework for holding the relevant actors accountable for addressing the emergence of DR-TB through research and treatment scale-up.
Inadequate funding for TB R&D
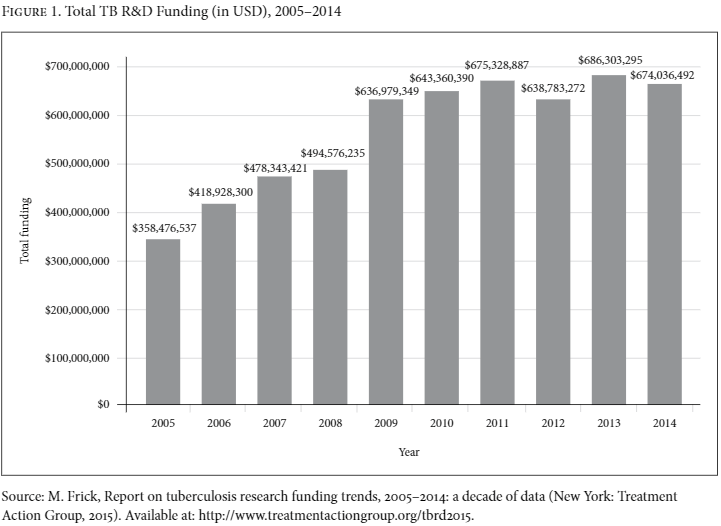
Over the past decade, funding for TB R&D has fallen far short of the resources required to improve TB treatment by developing new drugs and repurposing older ones. The Stop TB Partnership’s Global Plan to Stop TB, 2011–2015 estimates that the world needed to spend USD$9.8 billion between 2011 and 2015 to develop the new tools we need to eliminate TB.18 This covers spending across six areas of TB research: basic science, diagnostics, drugs, vaccines, operational research/epidemiology, and infrastructure. To create accountability toward this goal, Treatment Action Group (TAG) has tracked global spending on TB R&D every year since 2005 using a survey sent to funding organizations in the public, private, philanthropic, and multilateral sectors. (A detailed description of TAG’s resource-tracking methodology is published in the 2015 Report on Tuberculosis Research Funding Trends).19 Resource tracking conducted by TAG shows that by the end of 2014, the world had invested $2.7 billion in TB R&D, less than one-third of the $9.8 billion goal.
In each of the 10 years TAG has tracked TB R&D funding, annual spending on all forms of TB research by public, private, philanthropic, and multilateral institutions together has never exceeded $700 million. Alarmingly, funding for TB R&D appears to be flatlining. After increasing from $358.5 million in 2005 to $636.7 million in 2009, funding for TB R&D has remained stagnant since the global financial crisis (see Figure 1). As many observers have pointed out, flat funding is essentially falling funding, since inflation decreases the purchasing power of flat budgets.20 Moreover, the costs of biomedical research have risen faster than inflation, at least in the United States—the country that accounts for over half of all public spending for TB research.21 This underfunding is especially acute for TB drug research, which in 2014 received $243.3 million in funding, one-third of the Global Plan’s annual target of $740 million for TB drug R&D.22
In addition to its low absolute level, funding for TB R&D is highly concentrated among a small pool of donors, most of them public institutions in high-income nations. Sixty percent of TB research funding comes from public agencies, and 62% of public funding comes from a single country: the United States. Mirroring the global trend, US government funding for TB R&D has remained flat since 2009, when US government agencies spent $255.4 million on TB R&D. In 2014, the US government spent $248.5 million.23 This dependency on public funding from a small number of country governments is heightened by the high degree of concentration of available funding in other sectors. For example, the Gates Foundation gave 86% of philanthropic support for TB R&D in 2014, and Otsuka contributed 54% of pharmaceutical industry funding.24 This leaves public agencies with few industry and philanthropic partners, and has left the field vulnerable to shifting donor priorities and even the withdrawal of major funders.
This withdrawal has been most pronounced in the pharmaceutical sector. Since 2012, three major pharmaceutical companies (Pfizer, AstraZeneca, and Novartis) have closed their TB research programs as part of an industry-wide pivot away from anti-infectives research.25 (Although Novartis is renewing investment in the development of clofazimine for TB, it is not pursing new TB drug candidates.) The atrophying state of anti-infectives research has generated warning cries for over a decade.26 Newly developed antibiotics are not expected to generate blockbuster sales, leading major pharmaceutical companies to focus on developing new treatments for chronic illnesses or new biologics such as vaccines. In 2014, the pharmaceutical sector spent just $98.6 million on TB drug development, a decline of nearly one-third from 2011.27
As pharmaceutical companies pull back from anti-infectives and TB research, it will become imperative that governments act on their obligation to support research; inaction by states will not be met by compensatory efforts by actors from other sectors.
Inadequate access to TB treatment
Such a shallow and narrow funding pool would hinder progress against any disease, but is particularly ill-suited to respond to the challenges of treating TB in an age of increasing drug resistance. An estimated 480,000 people developed MDR-TB in 2013, but only 97,000 started treatment.28 While many of these people did not even receive a diagnosis, at least 39,000 patients (plus an unknown number detected in previous years) were on waiting lists to access treatment.29 For some, the wait to receive treatment resulted from availability issues tied to convoluted drug procurement practices, insufficient national TB program budgets, or drug shortages. For those in need of the novel drugs bedaquiline (developed by Janssen Therapeutics and approved by the US Food and Drug Administration in 2012) and delamanid (developed by Otsuka and approved by the European Medicines Agency in 2014), challenges to accessing treatment are even more complex and are partially attributable to the way in which TB research is financed, conducted, and owned.
Like medical research on many other diseases, most TB R&D occurs within a system in which ownership of innovation is governed by a “maximalist approach to intellectual property protection,” as defined by the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS).30 By setting required minimum levels of protection without specifying upper limits, TRIPS and related trade laws have set up a conflict between intellectual property (IP) protection and public health, with innovation caught in the middle.31 The current paradigm both grants inventors strong IP protections and establishes low barriers for securing effective monopolies, creating conditions which have allowed companies to price TB medicines unaffordably or fail to make them available altogether in non-lucrative markets by either choosing not to register products with national regulatory authorizes or by blocking generic manufacture.
To take one stark example, only 2% of the estimated 150,000 people each year who would benefit from bedaquiline or delamanid under current WHO guidance have received them.32 Access to companion drugs including linezolid and clofazimine, which are often required to build a regimen that is sufficiently robust to allow the use of bedaquiline or delamanid while minimizing the threat of acquired drug resistance, also remains poor. Clofazimine and linezolid were each developed to treat other bacterial infections and are currently used off-label to treat TB, which limits their availability for people with TB.33 High pricing, lack of registration in high MDR-TB burden countries, and slow movement of national TB programs to adopt new diagnostic and treatment strategies have all contributed to the poor uptake of these potentially life-saving drugs. Operational research studies using these drugs to treat extensively drug-resistant TB (XDR-TB) indicate that they can dramatically improve culture conversion, an early indication that treatment is working, from about 26% to 75%–97%, pointing to the grave costs of such a vast access gap.34
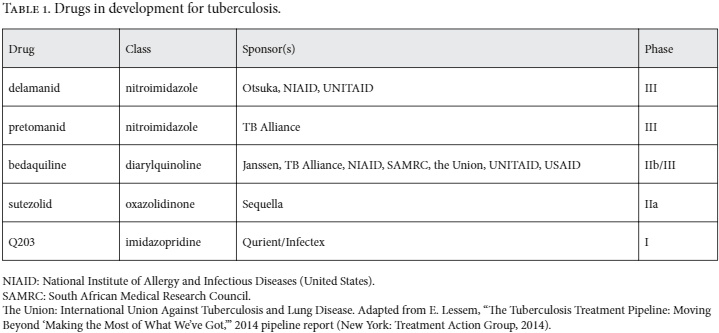
Other access challenges can be traced back to the ways in which inadequate funding has delayed the overall development of new drugs and forced compromises in specific drug research programs. The resulting tradeoffs do not always satisfy the conditions needed to create new regimens that would be patient friendly, clinically effective, and programmatically practicable.35 The impact of limited funding is evident in the small number and slow progress through the clinical pipeline of new drug candidates for TB. The FDA has only approved two new drugs for the treatment of TB since 1987, versus 36 drugs or drug combinations for HIV.36 Currently, there are only five new chemical entities in clinical trials for TB, from just four different drug classes, and only one candidate in Phase I trials (see Table 1).37 The number of candidates in Phase I serves as a bellwether for the health of an R&D pipeline, since to reach this phase, compounds must successfully traverse the so-called “valley of death” between laboratory studies in vitro and clinical trials in vivo.
The need for multiple drug candidates is especially great in TB, as treating TB requires combination therapy to protect against the development of resistance. Yet there are few new drugs to study, and even those have not yet been studied in combination.38 Instead, new TB drugs such as bedaquiline and delamanid were studied as add-ons to existing regimens, potentially improving the efficacy but doing nothing to address the toxicities, intolerabilities, drug interactions, and long duration of existing options. Bedaquiline and delamanid each received approval and reached the market with little to no data about the safety and efficacy of their use in combination with each other or with other new drugs still under development, and without evidence as to how they may optimize regimens repurposing older drugs.
Insufficient funding for TB R&D also adversely impacts the quantity and quality of science supporting the approval of new drugs, as clinical trials required for drug development are costly—estimates range from tens of millions to billions to bring a new drug to market.39 In TB trials, lengthy treatment times and the need for extensive follow-up to measure relapse-free cure add additional expenses. In 2014, the nearly $500 million funding gap for TB drug research was more than double the amount of available funding.40 This shortfall in funding for research translates into a paucity not only of new drug candidates, but also of trials informing the approval and use of the few new drugs that are developed. For example, delamanid received conditional approval for the treatment of MDR-TB from the EMA in 2014, based on data from six clinical trials—none of which was Phase III (the Phase III trial is currently underway, with results anticipated in 2018).41 By contrast, dolutegravir, a new drug approved by the EMA for the treatment of HIV in 2013, has been studied in 61 clinical trials.42
The combination of inadequate financing and a maximalist approach to IP protection means other promising compounds get stuck in early stages of development, and often remain unavailable for outside investigators to study. For example, sutezolid entered Phase I trials under Pfizer’s auspices in 2009. It then took four years for sutezolid to complete just three Phase I and Phase IIa trials—each under two weeks in duration—in which it showed promising early signs of safety and efficacy. Pfizer’s underinvestment in the candidate, then subsequent transfer of IP rights to private company Sequella (which asserts it cannot raise capital to develop the drug without receiving from Johns Hopkins University the rights to develop the drug in combination), have meant years of stagnation for one of very few promising TB drug candidates.43
As of March 2016, over two years after the Phase IIa trial ended, sutezolid has yet to begin Phase IIb trials.44 Other drugs of potential benefit go unstudied (for example, tedizolid, a recently approved drug to treat skin infections, which is also in the same class as sutezolid and likely has anti-TB activity). Many TB drugs are not even dose-optimized, including rifampicin—the backbone drug of the first-line TB treatment regimen, developed for TB in the 1950s—as well as moxifloxacin, pyrazinamide, and most second-line drugs for children.
Human rights and TB drug research and access
TB has direct implications for numerous human rights—including the rights to health, life, information, education, autonomy, participation, the benefits of scientific progress, and others. Analysis incorporating each of these rights can reveal ways to improve health systems’ responses to TB, and states are obligated to respect, protect, and fulfill these rights (that is, not directly violating rights; preventing rights violations by third parties; and ensuring policies, structures, and resources to realize rights).45 The three inadequacies discussed above—inadequate treatment options for TB, inadequate funding for TB R&D, and inadequate access to TB treatment—indicate that the current approach to addressing TB is insufficient, and each presents a compelling reason to focus on governments’ obligations under the rights to health and scientific progress, both as a practical means of improving state responses to TB and in furtherance of legal obligations under international human rights law. The following analysis focuses on these two rights in greater depth, but the interdependent nature of human rights, and the widespread implications of the TB epidemic, mean that the full framework of human rights is relevant.
Right to the highest attainable standard of health
The right to enjoy the highest attainable standard of health has been the focus of a considerable amount of guidance from human rights bodies and analysis in academic circles. States’ parties to the ICESCR have a binding legal obligation to take affirmative measures to ensure the right to health, including “the prevention, treatment, and control of epidemic […] diseases.”46 ICESCR Article 12 (2)(d) also requires states to take steps to ensure “[t]he creation of conditions which would assure to all medical service and medical attention in the event of sickness.”47 Understanding the extent of the legal obligation is important in this context. CESCR General Comment 14 on the right to health provides an authoritative elaboration and explanation of states’ obligations under Article 12.48 Like all human rights, the right to health imposes
three types or levels of obligations on States parties: the obligations to respect, protect and fulfil […]. The obligation to respect requires states to refrain from interfering directly or indirectly with the enjoyment of the right to health. The obligation to protect requires states to take measures that prevent third parties from interfering with article 12 guarantees. Finally, the obligation to fulfil requires states to adopt appropriate legislative, administrative, budgetary, judicial, promotional and other measures towards the full realization of the right to health.49
The right to health is not confined to health care, nor is it understood as a right to be healthy; rather, it encompasses inter alia the provision of conditions conducive to a healthy life, including underlying determinants of health (including adequate nutrition, sanitation, and information on health services).50
For diseases like TB, where inadequate and outdated tools hinder a vigorous public health response, fulfilling the right to health may require states to invest in research and development. General Comment 14 references medical research in several places, noting that fulfilling the right to health includes “the promotion of medical research and health education” and “fostering recognition of factors favoring positive health results, e.g., research and provision of information.” As described, the structure and level of TB R&D financing pose considerable challenges toward creating conditions conducive to a healthy life. As pharmaceutical companies scale back anti-infectives research, the responsibility will fall on governments—already the lead supporters of TB R&D—to increase activity in this area. This could translate to increased support for public sector research, or policies and programs that support R&D through partnerships with the private sector or nonprofit product developers. Regardless of the path chosen, states have an affirmative obligation to act on medical research as a necessary condition conducive to a healthy life.
The CESCR is unequivocal that “[t]he right to health in all its forms and at all levels contains the following interrelated and essential elements:” the availability, accessibility, acceptability, and quality of health facilities, goods, and services (3AQ).51 Taken in turn, each of the components under 3AQ demonstrates how the dearth of funding for TB R&D has created shortcomings in state progress toward upholding the right to health.
Availability
Functioning public health and health care facilities, goods, services, and programming must be available in sufficient quantity in a State party.52 On the short list of specifically enumerated requirements is the provision of essential drugs, as defined by the WHO Action Programme on Essential Drugs. TB medicines have long been required, and in 2014, WHO added five TB drugs to its Model List of Essential Medicines (EML): bedaquiline, delamanid, linezolid, terizidone, and rifapentine.53 In the press release announcing the list, WHO emphasized the preceding “45 years of scarce innovation for TB medicines” and called state attention to the hard work of ensuring the availability of these medicines.54 For many TB drugs, ensuring availability depends on first filling unmet research needs. Limited data on newly approved drugs have circumscribed their utility by leaving unanswered critical questions concerning safety and efficacy pending Phase III trials. In addition, vulnerable populations, including people with HIV, children, pregnant women, and people who use drugs, are underrepresented in TB drug trials, undermining the availability of optimal drug regimens for these groups.55
Accessibility
It is not enough for health facilities, goods, and services to simply be available—they must also be accessible to everyone.56 This includes nondiscrimination in the provision of treatment; that drugs are physically accessible to all; economically accessible (affordable); and that information on treatment and related health concerns is available. A key component of accessibility is the requirement that vulnerable and marginalized populations—often those hardest hit by TB—are able to access treatment. As above, better research into the optimal use of drugs and into their use in vulnerable populations is critical to ensuring nondiscrimination in access to and information on treatment. If adequately supported, TB R&D can aid in the accessibility of treatment and in the realization of the right to health for all. States may also need to structure support for TB research in ways that avoid the pitfalls of the current IP maximalist approach to innovation which has circumscribed access by allowing TB drug developers to develop new drugs singly rather than in combination; price new drugs out of reach; decide to not register products in non-lucrative markets; or restrict generic manufacture.
Acceptability
Health facilities, goods, and services must be respectful of medical ethics, culturally appropriate, sensitive to gender and lifecycle requirements, and designed to respect confidentiality and improve the health status of those concerned.57 The acceptability of current TB treatment options is jeopardized by the toxicities and side effects of current therapy that make it difficult for many people with TB to adhere to treatment. On top of general adherence challenges, current TB therapies are least acceptable to those most at risk from TB, including children, pregnant women, and people living with HIV.
Appropriately dosed pediatric formulations of first-line TB drugs did not exist until 2015, and research to establish the pharmacokinetic properties to determine optimal dosing of second-line TB drugs in children remains nascent.58 TB is one of the leading non-obstetric causes of death in pregnant women, yet pregnant women have been systematically excluded from TB drug research, even from trials where their safe inclusion would have been possible.59
Finally, many TB and HIV drugs have drug-drug interactions that either contraindicate their combined use or require dosing adjustments. Yet new TB drugs like bedaquiline reached the market with little data on their use with ARVs, and the majority of TB drugs trials have restricted the enrollment of people with HIV.60 Acceptability of TB treatment has been further reduced by the practice of studying new drugs as add-ons to existing inadequate regimens with little data about their safety and efficacy beyond the narrow combinations in which they were approved. The end result is treatment regimens that do not address the special needs of the groups most vulnerable to TB, a situation that can only be overcome by research to develop shorter, simpler, more tolerable regimens in doses and formulations acceptable to all people with TB.
State actors can rectify this acceptability gap by allocating funding for research in these special populations—the US National Institutes of Health’s (NIH) International Maternal Pediatric Adolescent AIDS Clinical Trials Network (IMPAACT) is one prominent example. States can also create regulatory requirements that ensure drug sponsors conduct such research: the EMA requires a pediatric investigational plan for new drugs, whereas the FDA could reverse its current exemption of standard pediatric study requirements for drugs for orphan diseases such as TB. Positive guidance, and even binding requirements, from regulators on including people with HIV and pregnant women in research could ensure that acceptability for vulnerable groups becomes the norm rather than the exception in TB treatment.
Quality
Health facilities, goods, and services must be scientifically and medically appropriate and of good quality. Scientifically approved, unexpired, and effective TB treatment regimens are critical, and increased support for TB drug R&D is required simply to remain at status quo; actual forward progress is the goal and would aid the advancement of the right to health. Quality of TB treatment would be improved by building a more robust evidence base to guide the use of old and new medicines. This would include research to increase the effectiveness of DR-TB treatment and eliminate the need to use drugs of poor quality in the sense of their limited efficacy and/or challenging toxicity and tolerability profiles. Once drugs are developed, research to increase the shelf life of TB drugs could have a considerable impact, particularly for low-volume but critical products.
Beyond the 3AQ, CESCR General Comment 14 highlights further access considerations in discussing the cornerstone principles of nondiscrimination and equality, asserting that “health facilities, goods and services must be accessible to all, especially the most vulnerable or marginalized sections of the population, in law and in fact, without discrimination on any of the prohibited grounds.”61 Equality of access to health care and health services is to be emphasized, and critically, “[i]nappropriate health resource allocation can lead to discrimination that may not be overt.”62 Vulnerable and marginalized populations are generally those most susceptible to TB, so better resource allocation and increased funding for TB drug R&D could help to address their needs, and reduce any discriminatory impact in conformity with the right to health.
Each of these observations underscores states’ obligations to support TB R&D under the right to health, but such observations must be tempered with the reality of resource constraints. A key concept that applies to state obligations for many economic, social, and cultural rights, including the right to health, is that of progressive realization.63 Essentially, progressive realization recognizes that states are at various stages of economic development, and that resource constraints may require a longer process towards fulfillment of certain rights. This does not obviate the obligations of States parties; the contrary is true—all States parties, regardless of resource constraints, must take affirmative steps towards the realization of rights, which includes meeting core obligations and ensuring non-discrimination. It does, however, mean that a state’s compliance with its obligations will be assessed in light of its available resources. Even in this context, it is notable that funding for TB drug R&D has remained stagnant in the face of inflation and despite the growing threat of DR-TB. Surveillance shows that TB incidence has been declining at the barely detectable rate of 2% per year; maintaining this pace would leave the world with a TB incidence rate in 2050 that is 1,000 times higher than the elimination threshold.64 In other words, economic and epidemiological trends suggest that states are not satisfying the conditions of progressive realization in their response to TB.
Even with the qualification of progressive realization, there is much room for state action on R&D. In the short term, states should support research to plug discrete holes in TB drug development, including improved pediatric formulations, dose optimization of backbone drugs like rifampicin, and capacity building for clinical trials. Longer-term endeavors could include increased support for TB basic science, new drug discovery initiatives, or Phase III clinical trials of new drug regimens. Each of these activities represents an affirmative step states could take to better the health of people with TB.
Right to enjoy the benefits of scientific progress and its applications
Attention to state obligations under the right to science further illuminates the ways in which current approaches to financing TB research undermine access to TB drugs and compromise the fight against TB. As articulated by ICESCR Article 15, States parties must recognize the right of everyone to: take part in cultural life (Article 15[a]); to enjoy the benefits of scientific progress and its applications (Article 15[b]); and to benefit from the material interests that stem from scientific, literary or artistic productions which one authors (Article 15[c]). Article 15 further specifies that full realization of the right requires that States parties take steps “necessary for the conservation, development and the diffusion of science and culture.”65
A plain language reading of the italicized sections above reveals that everyone has the right to enjoy the benefits of scientific progress and its applications, without discrimination, and that steps taken by states should include both the development and diffusion of science.66 It is significant that Article 15(2) singles out development and diffusion as distinct activities that exist on the same plane of concern for States parties. Article 15(1)(b) makes clear that the right encompasses not just scientific progress in the general sense of knowledge advancement and discovery, but the actual applications of this progress. In the case of TB research, state support for TB drug R&D would directly advance Article 15 (1)(b) and Article 15 (2), and increased access to treatment—particularly where access is currently limited—would aid in the fulfillment of this right through the diffusion of an important application of TB science: new and improved drugs.
Normative guidance from treaty bodies to date has focused on Article 15(1)(c) regarding intellectual property protection, and Article 15(1)(a) regarding the right to take part in cultural life.67 General Comment 17, for example, takes as its subject the author (the inventor or originator of research) and his/her material interests, rather than the end-user or intended beneficiary of research (the person with TB). CESCR has not yet issued a general comment on Article 15(1)(b), but discussion of its implications for funding research and ensuring access to the applications of scientific progress is underway.68 In 2010, the American Association for the Advancement of Science adopted a statement “On the human right to the benefits of scientific progress.”69 The statement notes that basic tenets of the right include “ensuring equitable access to the benefits of scientific progress, with a particular focus on vulnerable and marginalized groups” and “investing in R&D and creating incentives for innovation to address forms of suffering experienced by these groups.”
A 2012 report by the UN Special Rapporteur in the field of cultural rights that discussed the meaning and application of Article 15 pointed to four components of the right. One of these concerned “developing an enabling environment fostering the conservation, development and diffusion of science and technology.”70 Development
implies the adoption of programmes to support and strengthen publicly funded research, to develop partnerships with private enterprises and other actors […] and to promote freedom of scientific research. ‘Diffusion’ encompasses the dissemination of scientific knowledge and applications both within the scientific community and in society at large […] The diffusion of science is a precondition for public participation in decision-making and essential for fostering further research, development and applications.71
Further, speaking in the context of HIV—but with clear implications for TB—the UN Special Rapporteur in the field of cultural rights noted that the right to benefit from scientific progress is linked to several other rights including the right to health, and that
national case law can help to develop conceptual understanding of the right to science. In 2001, for example, the Supreme Court of Venezuela (Bolivarian Republic of) found that the failure of the Venezuelan Institute for Social Security to ensure a regular and consistent supply of the drugs needed by HIV-positive persons covered by it constituted, inter alia, a violation of the right to enjoy the benefits of scientific progress.72
Although normative guidance remains in development, states must still respect, protect, and fulfill the right through both development and diffusion.
Unfortunately, the current TB R&D funding system has hindered rather than enabled the development and diffusion of TB science. What measures would States parties need to enact to rectify this situation? Audrey Chapman has proposed a set of state functions that would uphold the obligation to fulfill the right to science. These functions include “setting priorities and channeling sufficient investment in a purposive development of science and technology,” particularly research bringing benefit to poor and disenfranchised groups; “providing opportunities for meaningful public engagement in decision-making about science and technology”; and the development of national plans of action with timetables, milestones and goals.73 Considered in turn, these suggestions point toward several ways states could intervene on TB research in accordance with fulfilling their obligations under right to science.
Public investment
As proposed by Chapman, “purposive investments” are those made by governments to address unmet scientific needs whose harms fall heaviest on marginalized groups and which non-public entities are unlikely to solve alone.74 TB research satisfies both conditions meriting a “purposive” public investment strategy. First, in funding TB research, governments would be addressing a disease that disproportionately affects poor and marginalized people, most of whom live in middle- and upper middle-income countries.75 Second, the sustained decline in private sector funding for TB research since 2011 means that the field’s reliance on government support will only intensify moving forward.
Complicating the issue of state support for research, however, is the fact that scientific research under the current IP maximalist system is increasingly privatized—even when funding may be primarily public in source. While most TB R&D funding comes from public institutions, industry groups have retained ownership over the trial data, patents, and marketing rights associated with most compounds in the TB drug pipeline. For example, the NIH and US Centers for Disease Control and Prevention (CDC) have funded nearly every candidate in the TB drug pipeline, most of which are owned by corporations.76 The disconnect between financing and ownership has resulted in the public paying twice for innovation—first to fund the research itself, and second to buy the resulting product for national TB programs at a price set by the company that benefited from public funding.77 In order to satisfy obligations under Article 15, purposive public investments in science must be accompanied by mechanisms that enable not just the development of new products like TB drugs, but also their diffusion.
States can draw on several tools to ensure that the applications of publicly funded science do not remain privately held and inaccessible. For example, governments could require patent pooling and the non-exclusive licensure of IP as conditions of receiving public funding. The potential of patent pooling to foster open, collaborative research would be maximized when paired with upfront grants (push funding) and prizes for meeting predetermined development milestones (pull funding). Médecins Sans Frontières (MSF) has proposed the combination of push, pull, and pool mechanisms under the so-called 3P Project as a way to incentive the development of new TB drug regimens.78 Regardless of the form the solution takes, the critical takeaway is that states, responsible for their legislative and regulatory schemes and committed to taking affirmative steps to increase compliance with human rights, should develop legislation, policy measures, and institutional mechanisms to ensure that individuals enjoy the benefits of scientific progress, even if scientific advancement is driven in part by the private sector.
Public participation
The inclusion of public voices in science and technology is also in keeping with the human rights principle of participation, which holds that all people have the right to participate in decision-making processes affecting their lives and well-being.79 While increasing public participation in science presents a formidable task, states face a relatively direct path of action in the case of TB R&D. TB activists have called for the meaningful engagement of TB-affected communities in the design, implementation, and dissemination of TB research.80 Much TB research advocacy has been directed by community advisory boards, groups that possess the scientific literacy required to participate in agenda setting with researchers.81 In addition, guidelines now exist to facilitate the engagement of communities in all stages of TB drug R&D.82
Public planning and accountability
Finally, the development of national plans of action for TB research with clearly delineated timetables, milestones, and goals would introduce an element of accountability that has been missing from previous promises made by states to support TB R&D (such as the 2009 Beijing Call for Action and the 2012 Delhi Communiqué).83 Countries need not tackle TB research or develop national action plans in isolation; WHO has announced plans to work with “pathfinder” countries to develop national strategic plans for TB research.84 States that develop such plans and put resources behind them would be acting in fulfillment of their obligations under the right to science. In the interest of inclusion, the development of such plans should include the engagement of a broad range of stakeholders, including members of TB-affected communities, providing further opportunity for states to create opportunities for participation in TB research in accordance with human rights standards.
Conclusion
TB is a preventable and curable disease that continues to claim over a million lives each year, due in large part to the limitations of current treatment options and the slow pace of TB research. In the context of stagnant or decreasing funding and a shift in private sector priorities away from TB R&D, governments—consistent with their legal obligations under the right to health and the right to science—can and should reassess the TB R&D system to improve the health and lives of their populations. In order to meet their obligation to fulfill the rights to health and scientific progress, governments will need to support TB R&D, and this may require actions that alter the ways TB research is currently financed, conducted, and owned, so that innovation is developed and diffused in a way that ensures all people with TB, their caregivers, and communities have access to TB research and its benefits.
Mike Frick, MSc, is the TB/HIV senior project officer at Treatment Action Group, New York, NY, USA.
Ian Henry, JD, is the legal fellow at the Program on Global Health and Human Rights at the USC Institute for Global Health, Los Angeles, CA, USA.
Erica Lessem, MPH, is the TB/HIV project director at Treatment Action Group, New York, NY, USA.
Contact info: Please address correspondence to the authors c/o Mike Frick, Treatment Action Group, 261 5th Ave, Suite 2110, New York, NY 11217. Email: mike.frick@treatmentactiongroup.org.
Competing interests: None declared.
Copyright © 2016 Frick, Henry, and Lessem. This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
References
- World Health Organization, Global tuberculosis report 2015 (Geneva: World Health Organization, 2015). Available at: http://www.who.int/tb/publications/global_report/en/.
- International Covenant on Economic, Social and Cultural Rights (ICESCR), G.A. Res. 2200A (XXI), Art. 12. (1966). Available at http://www2.ohchr.org/english/law/cescr.htm.
- L. Shaver, “The right to science and culture,” Wisconsin Law Review 1 (2010), pp. 121–184. Available at: http://papers.ssrn.com/sol3/papers.cfm?abstract_id=1354788.
- UN Committee on Economic, Social and Cultural Rights, General Comment No. 14, The right to the highest attainable standard of health, UN Doc. No. E/C.12/2000/4 (2000).
- Ibid.
- Shaver (see note 3).
- Universal Declaration of Human Rights (UDHR), G.A. Res. 217(A) (III) 1948, Art. 27. Available at: http://www.un.org/en/universal-declaration-human-rights/.
- Shaver (see note 4); A. Chapman, “Towards an understanding of the right to enjoy the benefits of scientific progress and its applications,” Journal of Human Rights 8/1 (2009), pp. 1–36.
- ICESCR Article 15 (see note 2).
- Ibid.
- Médecins Sans Frontières Access Campaign, “How many pills does it take?” Available at http://www.msfaccess.org/content/how-many-pills-does-it-take.
- World Health Organization, Global tuberculosis report 2014 (Geneva: World Health Organization, 2014). Available at http://www.who.int/tb/publications/global_report/en/.
- World Health Organization, Guidelines for the programmatic management of drug-resistant tuberculosis—2011 update (Geneva: World Health Organization, 2011. Available at http://www.who.int/tb/challenges/mdr/programmatic_guidelines_for_mdrtb/en/.
- S. Keshavjee and P. Farmer, “Tuberculosis, drug resistance, and the history of modern medicine,” New England Journal of Medicine 367/10 (2012), pp. 931–936.
- R. Upshur, J. Singh, and N. Ford, “Apocalypse or redemptions: responding to extensively drug-resistant tuberculosis,” Bulletin of the World Health Organization 87/6 (2009), pp. 481–483.
- S. Shah, “Majority of XDR-TB cases are due to transmission in a high HIV prevalence setting” (presentation at Conference on Retroviruses and Opportunistic Infections, Seattle, WA, February 23–26, 2015). Available at: http://www.croiconference.org/sessions/majority-xdr-tb-cases-are-due-transmission-high-hiv-prevalence-setting; S. Auld, “HIV-associated XDR-TB is transmitted in households and hospitals in South Africa” (presentation at Conference on Retroviruses and Opportunistic Infections, Boston, MA, February 22–25, 2016).
- K. Cohen, T. Abeel, A. McGuire, et al, “Evolution of extensively drug-resistant tuberculosis over four decades: whole genome sequencing and dating analysis of Mycobacterium tuberculosis isolates from KwaZulu-Natal,” PLoS Medicine 12/9 (2015), pp. e1001880.
- Stop TB Partnership, The global plan to stop TB, 2011–2015 (Geneva: World Health Organization, 2011). Available at http://www.stoptb.org/global/plan/.
- M. Frick, Report on tuberculosis research funding trends, 2005–2014: a decade of data (New York: Treatment Action Group, 2015). Available at http://www.treatmentactiongroup.org/tbrd2015.
- “Don’t hide the decline,” Nature 507/7491 (2014), pp. 139.
- amfAR and Treatment Action Group, The costs flat funding for biomedical research (Washington, DC: amfAR, The Foundation for AIDS Research, 2013). Available at http://www.amfar.org/articles/on-the-hill/2013/issue-brief–the-costs-of-flat-funding-for-biomedical-research/; Department of Health & Human Services, Biomedical research and development price index (BRDPI): fiscal year 2013 update and projects for FY 2014-FY 2019 (January 2014). Available at http://officeofbudget.od.nih.gov/pdfs/FY15/BRDPI_Proj_Jan_2014_508.pdf.
- Frick (see note 19).
- Ibid.
- Ibid.
- V. Dandekar, “Pfizer draws curtain on anti-infective research; refocuses on next-generation vaccines,” PharmAsia News (May 13, 2013). Available at https://www.pharmamedtechbi.com/publications/pharmasia-news/2013/5/13/pfizer-draws-curtain-on-antiinfective-research-refocuses-on-nextgen-vaccines; E. Mullin, “Global tuberculosis R&D takes hit with AstraZeneca site closure,” FierceBiotech (February 24, 2014). Available at http://www.fiercebiotechresearch.com/story/global-tuberculosis-rd-takes-hit-astrazeneca-site-closure/2014-02-24; C. Copley, “Novartis hands over experimental TB drugs in antibiotic pullback,” Reuters (August 20, 2014). Available at http://www.reuters.com/article/2014/08/20/us-novartis-tuberculosis-idUSKBN0GK19J20140820.
- See, for example, C. Nathan, “Antibiotics at the crossroads,” Nature 431/7011 (2004), pp. 899–902.
- Frick (see note 19).
- World Health Organization (see note 1).
- Ibid.
- Shaver (see note 3).
- Ibid; and A. Kapczynski, “The access to knowledge mobilization and the new politics of intellectual property,” Yale Law Journal 117/5 (2007), pp. 804–885.
- Médicins sans Frontières Access Campaign, DR-TB drugs under the microscope—4th edition (Geneva: Médicins sans Frontières, 2016). Available at http://www.msfaccess.org/sites/default/files/TB_report__DR-TB_DRUGS_UTM__4th_edition_2016.pdf
- Ibid.
- Ibid.
- G. Brigden, B. Nyang’wa, P. du Cros, et al, “Principles for designing future regimens for multidrug-resistant tuberculosis,” Bulletin of the World Health Organization 92/1 (2014), pp. 68–74.
- P. Clayden and M. Harrington, “Executive summary,” in A. Benzacar (ed), 2014 pipeline report (New York: Treatment Action Group, 2014), pp. 1–18. Available at http://www.pipelinereport.org.
- E. Lessem, “The tuberculosis treatment pipeline,” in A. Benzacar (ed), 2016 pipeline report [in press]. (New York: Treatment Action Group, 2016).
- Brigden (see note 35).
- Drugs for Neglected Diseases Initiative, An innovative approach to R&D for neglected patients (Geneva: DNDi, 2014). Available at: http://www.dndi.org/wp-content/uploads/2009/03/DNDi_Modelpaper_2013.pdf; and J. DiMasi, H. Grabowski, and R. Hansen, “Innovation in the pharmaceutical industry: new estiamtes of R&D costs,” Journal of Health Economics 47 (2016), pp. 20–33.
- Frick (see note 19).
- Otsuka, “Otsuka wins European marketing authorization for Deltyba™ (delamanid)” (April 30, 2014). Available at http://www.otsuka.co.jp/en/company/release/2014/0430_01.html. (Accessed 2014 May 2)
- European Medicines Agency, “Tivicay (dolutegravir) EPAR summary for the public,” (January 2014). Available at http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Summary_for_the_public/human/002753/WC500160681.pdf ; Clayden (see note 36).
- E. Lessem, “The tuberculosis treatment pipeline: moving beyond ‘making the most of what we’ve got,’” in A. Benzacar (ed), 2015 pipeline report (New York: Treatment Action Group, 2015), pp. 121–136. Available at: http://www.pipelinereport.org.
- National Library of Medicine, “NCT00990990 Safety, tolerability, pharmacokinetics and measurement of whole blood activity of PNU-100480 after multiple oral doses in healthy adult volunteers,” (Bethesda, MD: National Library of Medicine, August 2013). Available at https://clinicaltrials.gov/ct2/results?term=sutezolid&Search=Search.
- See, for example, D. Tarantola and S. Gruskin, “Human Rights Approach to Public Health Policy”, Health and Human Rights in a Changing World (New York: Routledge, 2013) pp. 44-45.
- ICESCR Art. 12 (see note 2).
- Ibid.
- CESCR General Comment No. 14 (see note 4).
- CESCR General Comment No. 14, para. 33 (see note 4).
- Ibid, 22.
- Ibid, 12(a).
- Ibid.
- World Health Organization, 19th WHO Model List of Essential Medicines (Geneva: World Health Organization, 2015). Available at http://www.who.int/medicines/publications/essentialmedicines/EML2015_8-May-2015.pdf.
- World Health Organization, “WHO moves to improve access to lifesaving medicines for hepatitis C, drug-resistant TB and cancers,” May 8, 2015. Available at http://www.who.int/mediacentre/news/releases/2015/new-essential-medicines-list/en/.
- H. McIlleron, S. Abdel Rahman, J. Dave, M. Blockman, A. Owen, “Special populations and pharmacogenetic issues in tuberculosis drug development and clinical research,” Journal of Infectious Diseases 211/S3 (2015), pp. S115–25.
- CESCR (see note 4).
- Ibid.
- L. McKenna, “Momentum in the pediatric tuberculosis treatment pipeline,” in A. Benzacar (ed), 2015 pipeline report (New York: Treatment Action Group, 2015), pp. 137–152. Available at http://www.pipelinereport.org.
- A. Gupta, J. Mathad, S. Abdel-Rahman, et al, “Toward earlier inclusion of pregnant and postpartum women in tuberculosis drug trials: consensus statements from an international expert panel,” Clinical Infectious Diseases 62/6 (2016), pp. 761–9.
- Lessem (see note 43).
- CESCR General Comment No. 14 para. 12 (see note 4)
- CESCR General Comment No. 14 para. 19 (see note 4)
- ICESCR Art. 2 para. 1 (see note 2)
- C. Dye, P. Glaziou, K. Floyd, and M. Raviglione, “Prospects for tuberculosis elimination,” Annual Review of Public Health 34 (2013), pp. 271–86.
- ICESCR Art. 15 (see note 2, emphasis added).
- United Nations, Vienna Convention on the Law of Treaties, United Nations Treaty Series Vol. 1155 (1969). Available at http://www.refworld.org/docid/3ae6b3a10.html.
- UN Committee on Economic, Social and Cultural Rights, General Comment No. 21, The right of everyone to take part in cultural life, UN Doc. No. E/C.12/GC/21.
- UN Committee on Economic, Social and Cultural Rights, General Comment No. 17, The right of everyone to benefit from the protection of the moral and material interests resulting from any scientific, literary or artistic production of which he or she is the author, UN Doc. No. E/C.12/GC/17 (2006).
- American Association for the Advancement of Science Board of Directors, “AAAS statement on the human right to the benefits of scientific progress,” April 16, 2010. Available at http://www.aaas.org/sites/default/files/content_files/UNReportAAAS.pdf
- Farida Shaheed, UN Special Rapporteur in the field of cultural rights, The right to enjoy the benefits of scientific progress and its applications, UN Doc. No. A/HRC/20/26. Available at http://www.ohchr.org/Documents/HRBodies/HRCouncil/RegularSession/Session20/A-HRC-20-26_en.pdf, p. 8.
- Ibid.
- Ibid.
- Chapman (see note 8).
- Ibid.
- World Health Organization (see note 1).
- Frick (see note 19).
- See, for example, L. McKenna, “Punk’d by pharma: public funds for private products,” TAGline 21/1 (2014), pp. 14–15. Available at http://www.treatmentactiongroup.org/tagline/2014/spring/punked-pharma-public-funds-private-products; and M. Balasegaram, “Drugs for the poor, drugs for the rich: why the current R&D model doesn’t deliver,” (BMJ, February 13, 2014). Available at http://blogs.bmj.com/bmj/2014/02/13/manica-balasegaram-drugs-for-the-poor-drugs-for-the-rich-why-the-current-research-and-development-model-doesnt-deliver/.
- Médecins Sans Frontières, “Push, pull, pool: accelerating innovation and access to medicines for tuberculosis,” November 7, 2013. Available at http://www.msfaccess.org/3Ps-project.
- http://www.unfpa.org/resources/human-rights-principles
- A. DeLuca, E. Lessem, D. Wegener, et al, “The evolving role of advocacy in tuberculosis,” Lancet Respiratory Medicine 2/4 (2014), pp. 258–9; and R. Boulanger, S. Seidel, E. Lessem, et al, “Engaging communities in tuberculosis research,” Lancet Infectious Diseases 13/6 (2013), pp. 540–545.
- DeLuca (see note 80).
- Critical Path to TB Drug Regimens, Good participatory practice guidelines for TB drug trials (Washington, D.C.: CPTR, 2012). Available at http://www.cptrinitiative.org/downloads/resources/GPP-TB%20Oct1%202012%20FINAL.pdf.
- The Delhi Communiqué (2nd BRICS Health Ministers’ Meeting, New Delhi, India, January 11, 2013). Available at http://www.brics.utoronto.ca/docs/130111-health.html; The Beijing “Call for Action” on Tuberculosis Control and Patient Care (Ministerial Meeting of High M/XDR-TB Burden Countries, Beijing, China, 1–3 April, 2009). Available at http://www.who.int/tb_beijingmeeting/en/.
- World Health Organization, Global Consultation on Research for TB Elimination (Stockholm: Karolinska Institute, 2014). Available at http://www.who.int/tb/features_archive/researchforTBelimination_meeting/en/.