Traditional/alternative medicines and the right to health: Key elements for a convention on global health
Emmanuel Kabengele Mpinga, Tshimungu Kandolo, Henk Verloo, Ngoyi K. Zacharie Bukonda, Ngianga-Bakwin Kandala, Philippe Chastonay
Health and Human Rights 15/1
Published June 2013
Abstract
Little has been done to investigate and promote the importance of non-conventional medicines (NCMs) in the realization of the right to health, yet all over the world people regularly resort to NCMs to secure healing or to prevent or mitigate the occurrence of a wide range of morbidities. This study aims to elucidate the theoretical framework of the role of NCMs in realizing the right to health, to identify the potential manifestations and causes of violations of the right to health in their practice, and to propose the practice of NCMs that could be included in a Framework Convention on Global Health.
We use both the documentary analysis and the violation of rights approaches. Through a non-directive review of the literature, we have tried to clarify the concepts and uniqueness of NCMs. We have also tried to unveil the challenges facing NCMs in a context where conventional medicines assume extensive power. The human rights approach has enabled us to bring to light the potential challenges to the rights of the various stakeholders that NCMs create.
We argue that NCMs can contribute to realizing the right to health through their availability, accessibility, acceptability, and relative quality. The Framework Convention on Global Health could contribute to the effective realization of this right by integrating basic principles to ensure the recognition, protection, promotion, and conservation of NCMs—at least of those NCMs that have shown evidence of efficacy—as well as catalyzing increased international cooperation in this area.
Introduction
Regardless of the various labels under which they are known, such as “traditional,” “complementary,” “alternative,” “integrative,” or “natural,” non-conventional medicines represent the primitive foundation of any health system. Societies have used them through the millennia for the maintenance of health.1
The knowledge, products, and practices stemming from these non-conventional medicines (NCMs) began to receive some international recognition during the 1970s under the International Drug Monitoring program of the World Health Organization (WHO).2 Interest in these medicines grew with various developments. In 2002, WHO adopted a world strategy to facilitate the integration of traditional medicines into health systems. This initiative was followed by political mobilization through new training programs in faculties of medicine, centers of research, and international meetings. Africa even established an annual day dedicated to these forms of medicine.3-6
These developments are occurring in a global health context marked by new health challenges that call for more effective organization in the health sector. Prominent among these challenges are the HIV pandemic, non-contagious diseases, and malnutrition.7 It must be stressed that approximately one billion persons have no access to modern health care. This lack of access is a primary cause of death around the world.8
In this context, one also must take into account the emergence of the human rights movement. Despite growing literature on clinical, ethical, and economic issues surrounding NCMs, research on the contribution of NCMs to the realization of the right to health is scarce.
Therefore, our study aims first to investigate the theoretical framework of the role of NCMs in the realization of the right to health, second to identify ways that the practice of NCMs may infringe the right to health, and third to suggest some general principles on NCMs that could be integrated into the proposal for a Framework Convention on Global Health (FCGH).
Methods
We used two approaches, namely the documentary analysis approach and the violation of rights approach. The documentary analysis involved a non-systematic review of the literature to clarify the concepts, to determine the degree of NCM use, and to assess the challenges they face. The violation of rights approach allowed us to identify potential risks of NCM stakeholder rights violations, thus setting milestones to be considered in a potential review of an FCGH.9-11
The study is organized into three sections. The first section seeks to clarify concepts of traditional and alternative medicines through a discussion about their respective definitions and the use of various forms of these medicines around the world.
The second section is essentially analytical. It considers the contributions of NCMs to the realization of the right to health. We discuss possible synergies and potential infringements on human rights generally and in the right to health specifically in the practice of these medicines.
The third section addresses fundamental principles related to NCMs that should be integrated into an FCGH. Indeed, the use of NCMs as tools alongside modern medicine for promoting the right to health might help resolve emerging global health challenges.
We conclude with the potential benefit of and protective role played by NCMs, and discuss their possible full recognition and protection as a strategy, along with modern medicine, for promoting the fulfillment of the right to health.
Traditional, alternative, additional, natural medicines: Concepts, appeal, stakes, and challenges
From Chinese medicine, to South African use of animal bones to treat disease, to Ayurvedic practices in India, so-called NCMs appear as complex social and cultural entities. They require a clarification of the theoretical framework, which supports an understanding of the motivations of their use.
Traditional, parallel, additional, alternative: From the monism to the pluralism of the health systems
The traditional attribute assigned to certain millennial practices of health prevention or health care refers to the past, to customs, beliefs, and legends. Thus, NCMs have often escaped any process of rationalization and empirical evidence.
There are several definitions of NCMs.12,13 The WHO definition is the most widely used, describing NCMs as “different practices, methods, knowledge and beliefs in health which imply the utilization for medical purposes of plants, animal parts and minerals, spiritual therapies, techniques and manual exercises, applied either individually or in combination to look at, to diagnose and to prevent the diseases or to protect the health.”14
For some authors, traditional medicine differs from conventional medicine by relying on theories not based on scientific data upon which modern medicine is based, thus bypassing the scientific evidence-based paradigm.15
The attributes “additional” or “alternative” translate an epistemological discomfort on the identity of these medicines, which are perceived as complements or alternatives to the modern medicine. According to the National Center for Complementary and Alternative Medicine and Zollmann and Vickers, “Complementary and alternative medicine is a broad domain of healing resources that encompasses all health systems, modalities, and practices and their accompanying theories and beliefs, other than those intrinsic to the politically dominant health system of a particular society or culture in a given historical period. Complementary and Alternative Medicine includes all such practices and ideas defined by their users themselves as preventing or treating illness or promoting health and well-being.”16
Some authors understand alternative medicine as a corpus of theories, knowledge, and practices of care susceptible to supplant modern therapeutic methods when those methods fail to achieve their goal. The notion of alternative medicine seems of limited use in countries where traditional medicine is predominant and where the system of care is not organized on the Western mode.17
For Synderman and Well, the concept of integrative medicine is the combination of the best modern medicine and the alternative and additional medicines for which the modern medicine has scientific proofs and sufficient guarantees regarding their safety and efficiency.18
Some authors consider NCMs as an offer in the health sector, which allows drawing from various medical systems, and thus can be considered as an answer to the expectations of multicultural societies. Others favor integrating NCMs into conventional medicine, especially in cancer centers, geriatrics units, and palliative care units where the main objective is controlling pain and ensuring quality of life rather than healing.19 In those places, different health specialists such as medical specialists, nutritionists, psychologists, occupational therapists, and practitioners of traditional medicine work side by side.16 Magny et al. consider integrative medicine as an approach of the human being in a more “global” and “holistic” way.20
In the literature and in health practice, there seems to be a need to substitute the monolithic power of modern medicine with a medical pluralism based on the fact that conventional medicine is only one of the multiple paradigms for diagnosing and treating diseases. This is a reminder that world cultures have historically resorted to different paradigms of preventive and curative approaches of disease.21,22 Traditional medicine extends well beyond the medicines themselves to a broader scope of experiences, practices, and products, including herbal medicine, acupuncture, spiritual or mineral practices, manual therapies, and exercises.
The term non-conventional medicine offers the advantage of including all the types of medicines known under the labels of “traditional,” “complementary,” “alternative,” “integrative,” or “natural,” thus, it is the term we use here.
The use of non-conventional medicines in globalized societies
The use of NCMs is widely documented in the literature. Their use remains widespread in all regions of the world and is increasing in industrialized nations. In China, traditional preparations from plants represent 30-50% of total medicine consumption.40 In Ghana, Mali, Nigeria, and Zambia, the initial treatment for 60% of the children suffering from high fever due to malaria is the use of healing plants administered at home. WHO reports that 80% of the African population resorts to traditional medicines, and that in several African countries, traditional midwives attend most births.23,24
In Europe, North America, and other industrialized regions, more than 50% of the population sought help at least once from alternative or complementary medicine. In San Francisco, London, and South Africa, 75% of people living with HIV/AIDS seek help from traditional, additional, or parallel medicines. Meanwhile, 70% of Canadians have sought help at least once from NCMs. In Germany, 90% of people take a natural remedy at some point in their life. NCMs are commonly used in treatment of degenerative and chronic diseases such as obesity, insanities, diarrheas, cancers, chronic fatigue syndrome, chronic pains, diabetes, and renal diseases.25-29
What explains the wide use of these medicines in our societies? Three theories have been proposed:
- Dissatisfaction of patients who have experienced the downside of conventional medicines, finding them sometimes ineffective, impersonal, overly high-tech, and expensive;
- Personal control by patients who resort to alternative medicines because they find them less authoritarian and more participative;
- Philosophic congruence, that is, alternative medicines are attractive because of their compatibility with the spiritual or philosophic world’s representation of health and diseases.30
For others, the growing interest in NCMs can be attributed to several reasons, including technological, economic, cultural, and social reasons. Furthermore, the Internet facilitates access to NCMs.31
Though the reasons that explain the interest in NCMs may seem obvious in developed societies, they do not take into account the contexts of developing countries. In these countries, where the populations have little access to the services and programs of modern medicines, NCMs may constitute the only available and accessible health care services.2
Non-conventional medicines: Stakes and major challenges
The persistence and current use of NCMs is not harmless. The analysis of this domain reveals complex stakes of political, economic, security, cultural, and legal nature.
Politically, recourse to NCMs is an answer to the health needs of the populations, which points to the responsibilities of governments on one hand, and on the other hand, relates to power struggles between diverse modalities of social organization and interests of numerous actors in the health sector. These medicines long represented a form of colonial resistance, as well as a strategy to insure health care for all, especially rural populations.32
NCMs represent an economic reality. In South Africa, the trade in traditional medicines is estimated to be worth 2.9 billion rand ($US 320 million) per year, representing 5.6% of the national health budget. With 27 million consumers, the trade is widespread. There are at least 133,000 individuals employed in this trade, especially rural women for whom this can represent an income, which in term might have an indirect beneficial effect on their families’ health.33 In industrialized countries, NCMs are valued by consumers. In Australia, out-of-pocket expenditure is estimated at $AU 4.13 billion ($US 3.12 billion) per year. In the US, the estimates have ranged between $US 27.0 billion and $US 34.4 billion for out-of-pocket expenditures and in England, out-of-pocket expenditure for six of the most established NCMs therapies has been estimated at over GB£ 450 million.1,34
Such large sums of money invested in NCMs might undermine the right to health, diverting spending from more effective health care. Besides not necessarily improving health, NCMs might even harm patients, either because the NCMs are themselves harmful or because they discourage and delay patients from seeking more effective care. Yet some of these medicines can help solve health problems, while bringing other benefits (such as a heightened sense of autonomy and spiritual health) that we have discussed.35
NCMs are cultural products, vectors of knowledge, but also a form of connection among members of the society. NCMs are inheritances of various societies and so from a social perspective, deserve protection.
The use of NCMs also implicates various human rights, including:
- The right to life, by the conservation of the human lives;
- The right to health, by the access to diverse therapies, where they have demonstrated their therapeutic efficacy;
- The right to intellectual property, because they belong to the communities that have been using them for centuries;
- The right to work, due to the fact that they generate income for traditional medicine practitioners;
Cultural rights, because they belong to and shape the identity of the peoples.
The interplay of the various issues has generated obstacles that deny traditional medicines their role and limit their impact on global health. These challenges are multiple:
1. The challenge of the national and international recognition
Their integration in the definitions of policies, priorities, and allocation of resources is limited and varies considerably world-wide. Grace notes that “increasing endorsement of [complimentary and alternative medicine (CAM)] stands in contrast to the negative attitude towards the CAM workforce by some members of the medical and other health professions and by government policy makers.” Some health systems facilitate collaboration between modern medicine and traditional medicine. A study in Northern Europe shows that traditional medicine is available in half of Norwegian hospitals and a third of Danish hospitals, indicating a change of attitude toward these medicines. 36,37 In Cameroon, Hillenbrand notes that in the urban environment, most traditional practitioners collaborate with medical practitioners; others routinely refer certain cases to hospitals, and many traditional practitioners receive patients who come to them from the hospital, either because they are dissatisfied with the conventional treatment or because they cannot afford the prescribed medicine.
In summary, liberal collaboration between the two types of medicines could facilitate and encourage a transfer of patients between them. It also implies the acknowledgement of strengths and limitations of the two medicines and of political will for the integration of both systems.38
2. The challenge of the regulations for the protection of knowledge and practices
The rules of protection included in international standards, which are applicable to the rights of patients, are currently not sufficient enough to protect traditional knowledge and biodiversity. The efforts of the World Intellectual Property Organization to identify the needs of traditional knowledge holders and to formulate principles for their protection resulted in no binding regulations. Thus the elaboration of guidelines for this purpose depends on the adoption of national laws in each state. Meanwhile, the Convention on Biological Diversity only considers NCMs in its genetic components, though these NCMs also have spiritual, psychological, or mineral components.9,41
3. The challenge of quality and possible toxicity
Products made with poor quality plants may constitute a serious threat to the safety of patients.23 For example, the plant Ma Huang (Ephedra), traditionally used in Chinese medicine against respiratory congestion, was marketed in the United States as a dietary aid and caused at least a dozen deaths through heart attacks or cerebrovascular accidents.42 In Belgium, at least 70 people had to undergo renal dialysis for an interstitial renal fibrosis after partaking in a diet prepared with erroneous plants.43 In certain regions of the word (South America, Asia, Africa) zoo-therapy is used as complementary medicine; 37 species of animals have been used in a simple treatment or in combinations with other drugs to treat asthma, which is not without danger since there are no scientifically based standards.44
4. The efficacy and effectiveness of the therapies
The question of the effectiveness of traditional medicines is central, given its implications for patient safety, financing, and public health in general. It is well debated. Vickers and other researchers maintain that NCMs cannot be evidence-based in the conventional sense, that “softer” types of evidence need to be taken into consideration as well, that placebo effect must not be dismissed as non-beneficial, and that the healing encounter includes factors that may never be quantifiable like hope or the power of belief.45,46 The scientific basis of NCMs is still questioned by many professionals all the more, an evaluation of the methodological qualities of randomized clinical trials (RCT) in NCMs shows that many RCT of complementary medicine interventions have relevant flaws.47
Some forms of NCMs have demonstrated efficacy through traditional scientific research methods. Randomized control trials have shown the benefits of diverse uses of acupuncture, certain healing plants, and certain manual therapies (physiotherapy, osteotherapy). For example, a meta-analysis of 29 randomized control trials of 17,992 people concludes that the global effects of acupuncture, as experienced by patients in clinical practice, are clinically relevant.48 However, due to missing, and even contradictory evidence, more research is needed on the efficacy of NCMs. So practitioners should be very careful not to harm patients when using NCMs.
5. Sustainability, bio-prospecting, and bio-piracy
If bio-prospecting can be understood as the “exploration, extraction and screening of biological diversity and indigenous knowledge for commercial value,” bio-piracy consists of the exploration of and the use for commercial purposes of genetic and biological resources, as well as traditional knowledge, without adequately compensating the local communities and states from which these resources arise. These practices, if not controlled, could bring about the extinction of endangered species and the destruction of resources and natural environments, thereby violating human rights.49,50
6. The challenges of valuing, training, promotion, and research in NCMs
Valuing traditional health knowledge and practices remains a subject of interest of several national, regional, or international authorities.3,4
In the field of training, for example, the situation varies worldwide. In 1998, 72% of US pharmacy schools offered instruction on healing plants.51 In South Korea, a survey from 2007 to 2010 showed that 85% of medical schools offered instruction on complementary or alternative medicines.52 In Europe, 40% of medical schools offered such teaching in 2006.53 In Africa, Akinola called for such instruction during basic medical training.54
All in all, the obvious paradox between a greater use of NCMs, the contribution of these medicines to the health of the individuals and the communities, the threats to the enormous safety and economic stakes that they represent, and the weaknesses of the existing mechanisms of regulation make the integration of NCMs in the FCGH imperative.
Non-conventional medicines and the right to health: Synergies and implications for the FCGH
Underestimated importance for the legal, operational, and technical plan
Though NCMs and the right to health both aim to ensure the best health conditions to all, the links between the two have neither been investigated nor taken into account in the definition and the implementation of health policies at national and global levels.
The right to health can be defined as an economic, social, and cultural right guaranteed by national and international legal instruments to ensure each individual the conservation of better health through actions aimed at improving social, political, and economic conditions of individuals and communities and a better functioning of the health care delivery system.
The main sources of this right at the international level are:
- The Universal Declaration of Human Rights (1948), Article 25;
- The Covenant on Economic, Social and Cultural Rights of 1966 Article 12, General Comment No. 14 of the Committee of the Economic, Social and Cultural Rights (August 11, 2000); other international legal instruments officially affirm the right to health, such the Convention on the Rights of Child 1989, the African Charter of Human Rights and the Peoples of 1981, the additional Protocol in the American Covenant on Human Rights dedicated to the economic, social and cultural rights of 1988, the European Social Charter of 1961 and its additional protocols, etc.
- International initiatives in the field of health or development contain global obligations for health. The objectives of the Millennium Development Goals constitute an illustration of the global awareness that health is one of the conditions of social development and reduction of poverty. Three of the eight objectives in this declaration pertain to health matters.55
- Finally, the mandates of several intergovernmental agencies entrust stakeholders with the responsibilities of realizing the right to health. It is the case for WHO, UNFPA, UNDP, the African Union, and the European Union.
The right to health is essential for the enjoyment of other rights but its realization also depends on the recognition and the effectiveness of other rights (for example, right to education, freedom of association, housing right). An analysis of the developments of the right to NCMs shows that the contribution and the role of these rights have hardly been taken into account, because:
- Article 12 of the International Covenant on the Economic, Social and Cultural Rights, which guarantees the right to health, contains no mention of the contribution of the NCMs. It also does not specifically mention conventional medicine or otherwise prescribe the types of medicine required to meet the highest attainable standard of health.
- In its General Comment No. 14 on the right to health, the Committee of Economic, Social and Cultural Rights takes a reductionist view by framing the question of NCMs as a matter of interest and concern only for native people. In doing this, the Committee misses what current data show, namely that everybody (including urban populations) resorts to non-conventional and complementary medicines. The Committee confines these medicines to the used materials (healing plants, animals, and minerals) without mentioning their psychological and spiritual components.56
- At the operational level, in his efforts to identify indicators of implementation of the right to health, the former special UN Rapporteur on the right to health underlines the necessity for health professionals to respect cultural differences. Yet a single reference mentions traditional practitioners among the healthcare professionals.57
- Finally, on the technical level, few health systems studies integrate the contributions of NCMs in matters of prevention or health care costs.
Obvious links for little-exploited synergies
The contribution of NCMs to the realization of the right to health has not been studied empirically. Yet a theoretical assessment of the comparative nature between the normative components of the right to health and the characteristics of NCMs might bring some clarification on possible links. Both approaches consider availability, accessibility, acceptability, quality of care, and non-discrimination as important features. This shows the potential of NCMs to contribute to the effective realization of the right to health where modern medicine falls short:
- Obligation to arrange sufficient installations, resources, services, and functional programs for public health and health care. This is a central obligation of states regarding the right to health. Many states are failing to fulfill these obligations. By contrast, NCMs are often readily available in these states, mostly without state investment. For example, in Ghana and Swaziland there are between 10,000 and 25,000 patients per each modern medical doctor versus 200 to 100 patients per traditional practitioner.3
- Obligation to make health installations, resources, services, and public health and health care programs accessible to all citizens without discrimination. This right to health obligation implies physical, economic, and cultural dimensions of accessibility. Affordability is often an obstacle to accessible formal health services. Yet NCMs often have low costs. A Japanese study of 1,000 patients indicated that the annual average cost of direct payment for the non-conventional medicines was one-half lower than that of modern medicines.58 However, this does not necessarily mean that the NCMs were effective.
- Obligation to make health care and public health infrastructure, resources, services, and programs readily acceptable to all users in terms of satisfaction with medical ethics and cultural sensitivity. This is a third right to health obligation, which emphasizes acceptability and requires health infrastructures and services to be delivered with respect and in a culturally acceptable manner. Cultural acceptance of NCMs, above all, explains their current use.30
- The demand for quality health care implies cultural acceptability, scientific rigor, and medical appropriateness of therapeutic procedures, services, technical installations, and properly qualified medical staff. In contrast to the possible advantages of NCMs cited above, certain NCM malpractice raises particular concerns. Charlatanism, exploitation of the patients, and unacceptable hygiene conditions are all too common, especially when these services escape or do not require the approval of competent authorities. Here, many traditional medicines show important flaws. Furthermore, therapeutic efficacy is a crucial issue: in this regard, NCMs have sometimes been shown to be therapeutically effective – but often not.
- Non-discrimination in the organization and supply of health services requires health infrastructure and services to be accessible to all, particularly to the most vulnerable and marginalized populations. Services must be administered according to the law, without discrimination by race, sex, age, and religion. In some regards, NCMs are less discriminatory since they are community-driven in developing countries and individually chosen in industrialized countries.
These links between NCMs and right to health are not unidirectional. The two areas offer significant possibilities of synergies, which generally have not yet been sufficiently exploited. The availability, accessibility, and quality of modern medicines do not seem to discourage the appeal of the NCMs or contribute to their disappearance. Establishing synergies between NCMs and modern medicines could improve global health care.15,59
Evidence of NCM effectiveness and the realization of the right to health
Evidence of the effectiveness of NCMs has been produced in certain contexts and for certain pathologies only. This is a crucial point, and it needs to be addressed by seeking scientifically rigorous studies for NCMs therapies that are similar to the current investigatory designs of modern medical therapies. A review of randomized control trials on traditional medicine that was published in JAMA evaluated the use of six alternative therapies for treatment of common clinical conditions. It found that chiropraxis spinal manipulation was not effective for episodic tension, Chinese herbal medicine formulation improved symptoms of irritable bowel syndrome, acupuncture was no more effective than amiptriptyline or placebo for relieving pain, and palmetto extracts improved urologic symptoms in patients with benign prostatic hyperplasia.
In oncology, a study carried out in 14 European countries on the use of NCMs by 956 cancer patients showed that the majority used NCMs to increase physical and emotional well-being, and that many seemed to have benefited from using NCMs. The authors recommended that health professionals explore the use of NCMs with their cancer patients.60,61
NCMs’ contribution to the right to health raises two other issues related to cost-effectiveness and state financing. NCMs therapies may be considered cost-effective compared to usual care for various conditions, including acupuncture for migraine, manual therapy for neck pain, spa therapy for Parkinson’s, relaxation therapy and potassium diet for cardiac patients. Authors of one analysis conclude that many NCMs have been shown to have good value and they call for further evaluation of the majority of NCMs.62
Regarding financing, reimbursement for many therapies is not automatic in countries with insurance systems. In many cases, reimbursement of NCMs is subject to the evidence of their therapeutic effectiveness, as it is the case in Switzerland for homeopathy, neural therapy, anthroposophical medicine, physiotherapy, and Chinese traditional medicine. 64
Where effectiveness is limited or nonexistent, devoting considerable state sums to supporting traditional medicines hinders rather than advances the full realization of the right to health. This is because states could have spent the funds in ways that would have more effectively advanced the highest attainable standard of health. States should spend the money in ways it will best advance health; thus, therapeutic efficacy is key. Currently, states misallocate funds. The 2001 Abuja Declaration commits all African Union member states to spend at least 15% of their national budgets on the health sector, however, in two-thirds of these countries spending is below 10%. Meanwhile, military spending in Sub-Saharan Africa has increased by 64% from 2001 to 2011.65
In Europe, health spending remained between 7.43% and 7.74% of GDP from 1998 to 2005, but during a similar period ( 2002 to 2011), military spending increased over 17%.66 Even larger increases in military spending have been reported from North America and Asia.67,68
One could speculate that allocating financial resources for the right to health might not be compromised by financing NCMs, and that funding related violations of the right to health develop primarily from iniquity, waste, and economic interests of governments. Nonetheless, financing for NCMs should not become an excuse not to fulfill other government obligations towards basic health care.
Traditional medicines might further contribute to public health, as they can lead to drug discoveries.62,63
Human rights violations in the practices of NCMs: Basic principles on the FCGH
The FCGH aims to improve the health of populations by more effectively realizing the right to health. We propose that the FCGH incorporate NCMs, since NCMs have potential benefits and have the potential to violate the rights of the various stakeholders involved in the practices of NCMs. 69,70
These stakeholders have different roles and functions, with divergent interests regarding NCMs and global health. Since, interactions between these stakeholders can result in infringements on rights, they warrant regulation by the FCGH (Figure 1).
1. The activity or inactivity of the state actors can lead to unchecked, unreliable, and unsafe practices for patients. Patients can be harmed if states fail to protect users or fail to reimburse certain therapies. State weaknesses can facilitate bio-piracy and the excessive exploitation of natural resources needed for NCMs.
2. Traditional practitioners can exercise their business illegally, without control by the state, and may fail to pay state taxes. They can violate patients’ privacy, jeopardize patients’ physical integrity, and fail to provide patients with information on a treatment’s side effects. Traditional practitioners may fail to refer patients, who would benefit from effective therapies (for example, those with tuberculosis or HIV), to conventional medicine services. Also, if they fail to collect information on diseases they observe, or they do not transmit this information to health authorities, they jeopardize the quality of health information systems.
3. Non-state actors, such as multinational companies, can extort traditional knowledge and secure patents for their own profit. They can take advantage of native peoples who were the original owners of that knowledge, without compensating them or respecting their intellectual property rights.
4. Patients seeking NCMs might infringe on the rights of others, for example by disrespecting the confidentiality and privacy of other patients.
This analysis suggests that the benefits and risks of NCMs exceed national boundaries and may be well served by enforcing an international regulatory framework that covers these interactions.
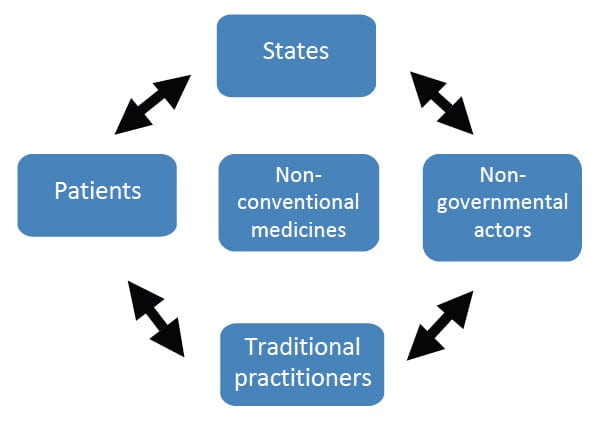
Figure 1: Key actors and their interactions with NCMs
Non-Conventional Medicines in an FCGH: Motivations and basic principles
Before articulating the basic principles that should be integrated into an FCGH, it is important to examine the nature and the extent of the existing regulations and their limits.
Legislation on the NCMs: World perspective
According to available estimates, only 25 of WHO’s 191 member states have any type of national policy on NCMs and alternative medicines, and only 64 countries regulate herbal medicines. From 1994 to 2001, the number of countries with regulations for healing plants increased from 50 to 70.71,72
On the international level, legal instruments that address NCMs include:
- The Declaration on the Cultural Diversity of UNESCO (2001)
- The Declaration of the United Nations on the Rights of Native (2007)
- The Declaration of the World Health Organization on the Traditional Medicines (2008)
- The Convention for the Protection of the Immaterial Cultural Heritage of UNESCO (2003)
- The Convention on Biological Diversity of United Nations (2003)
Furthermore, there are guidelines (such as those promulgated by WHO) dealing with:
- Quality control methods for medicinal plants (1998)
- Safety monitoring of herbal medicines in pharmacovigilance systems (2004)
- Regulation of herbal medicines in South East Asia Region (2003)
- Methodologies on research and evaluation of traditional medicines (2000)
- National policy on traditional medicines and regulation of herbal medicines (2005)
- Good agricultural and collection practices for medicinal plants (2003)71
Other UN agencies have issued guidelines that indirectly concern NCMs, such as the World Intellectual Property Organization’s protection of traditional knowledge, to a situation where NCMs traditional cultural expressions, and legislative texts concerning the genetic resources (expressions of the folklore).73
Furthermore some states, including members of the European Union, have issued directives in the field of NCMs, such as directive 2004/24/EC.74
Due to the fragmented nature of the regulations, each national regulatory authority approaches NCMs based on its own mandate and interests. These mandates and interests may be incomplete and may not be in harmony with the most effective NCMs policies and practices. Furthermore, surveillance of NCMs and the mechanisms used to remedy NCMs-related human rights violations are generally weak. This leads NCMs-related rights violations are common and leads us to believe that NCMs are ripe for inclusion in an FCGH.50
Basic principles of non-conventional medicines in the FCGH
The FCGH should incorporate several key points concerning NCMs.
Four key ideas on NCMs
- States parties in this Convention should:
- Recognize the importance of NCMs that have proven to be effective according to international scientific standards. NCMs contribution to the health of populations and the benefits they offer in terms of accessibility, availability, and cultural acceptability should also be recognized;
- Face challenges of NCMs such as recognition, organization, quality of their services, safety of patients, rights and duties of non-state actors, and conservation of resources and their piracy;
- Recognize the need to protect users against malpractice (side effects and failures) in NCMs and ensure that users have remedies when their rights have been violated;
- Commit to protect, promote, and value NCMs that have proven effective in improving people’s health, while simultaneously valuing their non-medical benefits, by adopting laws, creating structures, and implementing research programs. Also prioritize protection against poor quality and other potential harms.
Six major principles on NCMs in the FCGH
We suggest that the convention emphasize the following principles and specific measures pertaining to NCMs:
1. Recognition of NCMs. Under this principle, states would recognize the importance and role of the knowledge and practices of NCMs that have proved effective according to international scientific standards in health care, as well as their contribution to the development of communities. This recognition would become integrated into constitution, in the law of the country, or in the sanitary code of every state.
2. Protection and conservation of NCMs. Under this principle, states would commit to setting appropriate measures to protect the knowledge, resources, and rights and responsibilities of the various actors involved in NCMs (states, patients, practitioners, non-state actors). To fulfill this requirement, states could adopt specific laws on NCMs and issue related regulations. Alternatively, states could adopt a national strategy on NCMs as a constitutive part of their national health policies.
3. Promotion and the valorization of NCMs. Under this principle, states would monitor the development of NCMs that have proven effectiveness according to international scientific standards by introducing and by supporting research, training, and political and scientific visibility of NCMs. States would create national institutions of research on NCMs and would integrate elements of NCMs into health professionals’ training programs.
4. Protection against harms of NCMs. States should protect users against malpractice involving traditional medicines, and take necessary measures to protect users against toxicity, inefficiency, low quality, poor hygiene, and assertions of witchcraft. These recommendations should be integrated in national laws and policies.
5. Sustainable use of resources. The obligation to ensure sensitive and sustainable exploitation of NCMs sources should also be registered in the law. States would commit to using NCM resources in a rational and sensible way.
6. Cooperation in the field of NCMs. States would recognize the importance of cooperation in the field of NCMs. They would collaborate on specific projects and create and support regional NCMs centers and exchanges through scientific and economic agreements.
Conclusion
NCMs are the common heritage of the human species and the testimony of our capacity to face diseases endangering human lives. For much of the world’s population, they remain the first step of prevention and care.
But NCMs face numerous challenges, including questions involving their credibility, effectiveness, efficiency, safety, and the rights of patients and other stakeholders. The lack of regulations or weakness of regulations might be one of the reasons for this situation. NCMs’ credibility also suffers from their relative lack of proven effectiveness.
By developing and integrating basic principles aimed at the recognition, protection and conservation of these medicines, we can promote and value them, and provide a special focus on their therapeutic validity that can correspond to modern scientific standards. Integrating NCMs into the FCGH may contribute to the effective realization of the right to health.
Emmanuel Kabengele Mpinga, PhD, MHA, MPH, is Associate Professor SSPH+ of Health and Human Rights at the Institute of Social and Preventive Medicine, University of Geneva in Geneva, Switzerland.
Tshimungu Kandolo, PhD, MPH, is Associate Professor of Epidemiology and Public Health Sciences at the Institut Supérieur des Techniques Médicales in Kinshasa, Democratic Republic of Congo.
Henk Verloo, RN, MSc, is a PhD candidate and is Professor at the Applied University of Health Sciences La Source in Lausanne, Switzerland.
Ngoyi K. Zacharie Bukonda, PhD, MPH, is Professor of Public Health Sciences in the Department of Public Health Sciences at Wichita State University in Wichita, Kansas, USA.
Ngianga-Bakwin Kandala, PhD, is Principal Research Fellow (Associate Professor) in Health Technology Assessment at the University of Oxford (KEMRI-Oxford Wellcome Trust Collaborative Programme) in Oxford, UK and in the Division of Health Sciences at Warwick Medical School in Coventry, UK.
Philippe Chastonay, MD, MPH, is Associate Professor of Public Health at the Institute of Social and Preventive Medicine and the Unit of Development and Research in Medical Education, University of Geneva in Geneva, Switzerland.
Please address correspondence to the authors c/o Emmanuel Kabengele Mpinga, email: Emmanuel.Kabengele@unige.ch.
References
1. G. Brodeker and F. Kronenberg, “A public health agenda for traditional, complementary and alternatives medicines,” American Journal of Public Health 92/10 (2002), pp. 1582-1591.
2. WHO, Guidelines on safety monitoring in herbal medicines in pharmacovigilance systems (Geneva: WHO, 2004).
3. WHO, Traditional medicine strategy 2002-2005 (Geneva: WHO, 2002).
4. Pan American Health Organization, Traditional complementary and alternatives medicines and therapies: Evaluation of plan of work 2001-2002, and plan of work 2002-2003 (Washington, D.C.: PAHO, 2003).
5. J. J. Brokaw, G. Tunnicliff, B. U. Raess, and D. W. Saxon, “The teaching of complementary and alternative medicine in U.S. medical schools: A survey of course directors,” Academic Medicine 77/9 (2002), pp. 876-881.
6. G. L. Sambo, “The decade of African traditional medicine: Progress so far,” African Health Monitor 13 (2010), pp. 4-6.
7. A. Shah, “Health issues,” Global Issues (September 22, 2011). Available at http://www.globalissues.org/issue/587/health-issues.
8. WHO, “Inequalities are killing people on grand scale, reports WHO’s commission,” WHO Press Release (August 28, 2008). Available at http://www.who.int/mediacentre/news/releases/2008/pr29/en/index.html.
9. A. R. Chapman, “A ‘violations approach’ for monitoring the International Covenant on Economic, Social and Cultural Rights,” Human Rights Quarterly 18/1 (1996), pp. 23-66.
10. Ajazuddin and S. Saraf, “Legal regulations of complementary and alternative medicines in different countries,” Pharmacognosy Review 6/12 (2012), pp. 154–160.
11. A. Buyse, “Lost and regained?: Restitution as a remedy for human rights violations in the context of international law,” Heidelberg Journal of International Law 68 (2008), pp. 129-153.
12. O. Caspi and O. Baranovitch, “When science meets medical tradition: What is needed for a dialogue on integrative medicine?” Journal of Alternative and Complementary Medicine 15/5 (2009), pp. 579-583.
13. O. Caspi, L. Sechrest, H. C. Pitluk, et al., “On the definition of complementary, alternative, and integrative medicine: Societal mega-stereotypes vs. the patients’ perspectives,” Alternative Therapies in Health and Medicine 9/6 (2003), pp. 58-62.
14. WHO, General guidelines for methodologies on research and evaluation of traditional medicine (Geneva: WHO, 2000).
15. X. F. Yan, Q. Ni, J. P. Wei, and H. Xu, “Evidence-based practice method of integrative Chinese and Western medicine based on literature retrieval through PICO question and complementary and alternative medicine topics,” Chinese Journal of Integrative Medicine 16/6 (2010), pp. 542-548.
16. C. Zollman and A. Vickers, “What is complementary medicine,” British Medical Journal 319 (1999), pp. 693-696.
17. F. Kronenberg, P. Molholt, M. L. Zeng and D. Eskinazi, “A comprehensive information resource on traditional, complementary, and alternative medicine: Toward an international collaboration,” Journal of Alternative and Complementary Medicine 7/6 (2001), pp. 723-729.
18. R. Snyderman and A. Weil, “Integrative medicine: Bringing medicine back to its roots,” Archives of Internal Medicine 162 (2002), pp. 395–397.
19. J. H. Flaherty and R. Takahashi, “The use of complementary and alternative medical therapies among older persons around the world,” Clinics in Geriatric Medicine 20/2 (2004), p. 179.
20. J. C. Magny, G. Harvey, Y. Levesque, et al. Pour une approche intégrée en santé (Quebec: Presses de l’Université du Québec, 2009).
21. Y. C. Huang, I. H. Lin and M. L. Yeh, “Developments and trends in traditional medicine and complementary and alternative medicine,” Hu Li Zha Zhi Journal of Nursing 55/2 (2008), pp. 81-85.
22. J. L. Hamilton, B. Roemheld-Hamm, D. M. Young, et al., “Complementary and alternative medicine in US family medicine practices: A pilot qualitatitive study,” Alternative Therapies in Health and Medicine 14/3 (2008), pp. 22-27.
23. J. Segar, “Complementary and alternative medicine: Exploring the gap between evidence and usage,” Health: An Interdisciplinary Journal for the Social Study of Health, Illness and Medicine 16/4 (2012), pp. 366-381.
24. M. K. Lim, P. Sadarangani, H. L. Chan and J. Y. Heng, “Complementary and alternative medicine use in multiracial Singapore,” Complementary Therapies in Medicine 13/1 (2005), pp. 16-24.
25. J. M. Shim, G. Bodeker, and G. Burford, “Institutional heterogeneity in globalization: Co-development of western-allopathic medicine and traditional-alternative medicine,” International Sociology 26/6 (2011), pp. 769-788.
26. K. C. Chinsembu and M. Hedimbi, “An ethnobotanical survey of plants used to manage HIV/AIDS opportunistic infections in Katima Mulilo, Caprivi region, Namibia,” Journal of Ethnobiology and Ethno medicine 6/25 (2010).
27. H. Boon, ”Regulation of complementary/alternative medicine: A Canadian perspective,” Complementary Therapies in Medicine 10/1 (2002), pp. 14-19.
28. R. Stange, R. Amhof, and S. Moebus, “Complementary and alternative medicine: Attitudes and patterns of use by German physicians in a national survey,” Journal of Alternative and Complementary Medicine 14/10 (2008), pp. 1255-1261.
29. G. N. Mbeh, R. Edwards, G. Ngufor, et al., “Traditional healers and diabetes: Results from a pilot project to train traditional healers to provide health education and appropriate health care practices for diabetes patients in Cameroon,” Global Health Promotion 17 /2 (2010), pp. 17-26.
30. J. A. Astin, “Why patients use alternative medicines: Results of a national study,” Journal of the American Medical Association 279/19 (1998), pp. 1548-1553.
31. T. Passarelli, Complementary and alternative medicine in the United States (2008). Available at http://www.cwru.edu/med/epidbio/mphp439/complimentary_meds.pdf.
32. A. Wahlberg, “Bio-politics and the promotion of traditional herbal medicine in Vietnam,” Health 10/2 (2006), pp. 123-147.
33. M. Mander, L. Ntuli, N. Diederics and K. Mavundla, “Economics of traditional medicine trade in South Africa,” in S. Harrison, R. Bhana, and A. Ntuli (eds), South African Health Review (South Africa: Health Systems Trust, 2007).
34. J. Spinks and B. Hollingsworth, “Are the economics of complementary and alternative medicine different to conventional medicine?” Expert Review of Pharmacoeconomics & Outcomes Research 9/1 (2009), pp. 1-4.
35. W.B. Jonas, “Directions for research in complementary medicine and bioterrorism,” Alternative Therapies in Health and Medicine 8/2 (2002), pp. 30-31.
36. S. Grace, “CAM practitioners in the Australian health workforce: An underutilized resource,” BMC Complementary and Alternative Medicine 12/205 (2012).
37. L. J. Salomonsen, L. Skovgaard, S. la cour, et al., “Use of complementary and alternative medicine at Norwegian and Danish hospitals,” BMC Complementary and Alternative Medicine 11/4 (2011).
38. E. Hillenbrand. “Improving traditional-conventional medicine collaboration: Perspectives from Cameroonian traditional practitioners,” Nordic Journal of African Studies 15/1 (2006), pp. 1-15.
39. WHO, National policy on traditional medicine and regulations of herbal medicines (Geneva: WHO, 2005).
40. World Intellectual Property Organization, Intellectual property needs and expectations of traditional knowledge holders (Geneva: WIPO, 2001).
41. United Nations, Convention on Biological Diversity (1992).
42. T. Efferth and B. Kaina, “Toxicities by herbal medicines with emphasis to traditional Chinese medicine [Review],” Current Drug Metabolism 12/10 (2011), pp. 989-996.
43. C. Anquez-Traxler, “The legal and regulatory framework of herbal medicinal products in the European Union: A focus on the traditional herbal medicines category,” Drug Information Journal 45/1 (2011), pp. 15-23.
44. R. R. N. Alves, H. N. Lima, M. C. Tavares, et al., “Animal-based remedies as complementary medicines in Santa Cruz do Capibaribe, Brazil,” BMC Complementary and Alternative Medicine 8 (2008), p. 44.
45. A.Vickers, “Methodological issues in complementary and alternative medicine research: A personal reflection on 10 years of debate in the UK,” Journal of Alternative and Complementary Medicine 2/4 (1996), pp. 513-524.
46. E. Ernst, “Complementary medicine: Where is the evidence?” Journal of Family Practice 52/8 (2003), pp. 631-34.
47. K. Linde, W. B. Jonas, D. Melchart, and S. Willich, “The methodological quality of randomized controlled trials of homeopathy, herbal medicines and acupuncture,” International Journal of Epidemiology 30 (2001), pp. 526-531.
48. A. J. Vickers, A. M. Cronin, A. C. Maschino, et al., “Acupuncture for chronic pain: Individual patient data meta-analysis,” Archives of Internal Medicine 172/19 (2012), pp. 1444-1453; P. B. Fontanarosa and G. D. Lundberg, “Alternative medicine meets science,” Journal of the American Medical Association 280/18 (1998), pp.1618-1619.
49. P. Zakrzewski, “Bioprospecting or biopiracy?: The pharmaceutical industry’s use of indigenous medicinal plants as a source of potential drug candidates,” Complementary and Alternative Medicine 79/3 (2002), pp. 252-254.
50. L. Laursen, “Monsanto to face biopiracy charges in India,” Nature Biotechnology 30/1 (2012), p. 11.
51. D. M. Rowell and D. J. Kroll, “Complementary and alternative medicine education in United States pharmacy schools,” American Journal of pharmaceutical education 62 (1998), pp. 412-419.
52. D. Y. Kim, W. B. Park, H. C. Kang, et al., “Complementary and alternative medicine in the undergraduate medical curriculum: A survey of Korean medical schools,” Journal of Alternative and Complementary Medicine 18/9 (2012), pp. 870-874.
53. O. Varg, S. Marton and P. Molnar, “Status of complementary and alternative medicine in European medical schools,” Forsch Komplementmed 13 (2006), pp. 41-45.
54. B. O. Akinola, “Should complementary and alternative medicines familiarisation modules be taught in African medical schools,” Journal of Chinese Integrative Medicine 9/11(2011), pp. 1165-1169.
55. WHO, Keeping the promises: Health and the millennium development goals (Geneva: WHO, 2005).
56. Committee on Economic, Social and Cultural Rights, General Comment No. 14, The Right to the Highest Attainable Standard of Health, UN Doc No. E/C.12/2000/4 (2000). Available at http://www.unhchr.ch/tbs/doc.nsf/0/40d009901358b0e2c1256915005090be?Opendocument.
57. G. Backman, P. Hunt, R. Khosl, et al., “Health systems and the right to health: An assessment of 194 countries,” Lancet 372/9655 (2008), pp. 2047–2085.
58. H. Yamashita, H. Tsukayamaa, and C. Sugishitab, “Popularity of complementary and alternative medicine in Japan: Telephone survey,” Complementary Therapies in Medicine 10/2 (2002), pp. 84–93.
59. T. J. Kaptchuk and G. M. Franklin, “What is the best and most ethical model for the relationship between mainstream and alternative medicine: Opposition, integration, or pluralism?” Academic Medicine 80 /3(2005), pp. 286-290.
60. P. B. Fontanarosa and G. D. Lundberg, “Alternative medicine meets science,” Journal of the American Medical Association 280/18 (1998), pp. 1618-19.
61. A. Molassiotis, P. Fernadez-Ortega, D. Pud, et al., “Use of complementary and alternative medicine in cancer patients: A European survey,” Annals of Oncology 16 (2005), pp. 655–663, 2005.
62. P. M. Herman, B. M. Craig, and O. Caspi, “Is complementary and alternative medicine (CAM) cost effective? a systematic review,” BMC Complementary and Alternative Medicine 5/11 (2005).
63. B. Graz, A. Y. Kitwa, and H. M. Malebo, “To what extent can traditional medicine contribute a complementary or alternative solution to malaria control programmes?” Malaria Journal 10/Suppl. l1 (2011), p. S6.
64. Swiss Confederation, “Five complementary medicine therapies will be conditionally reimbursed for an interim period of six years” (December 1, 2011). Available at http://www.bag.admin.ch/aktuell/00718/01220/index.html?lang=fr&msg-id=37173.
65. Stockholm Peace Research Institute, Background paper on SIPIRI military expenditure data 2011 (Stockholm: SPRI, 2012).
66. WHO Europe, The European health report 2009: Health and health systems (Copenhagen: WHO, 2009); Stockholm Peace Research Institute (see note 65).
67. US Department of State, “Highlights and trends: Military expenditures,” World Military Expenditures and Arms Transfers (2005). Available at http://www.state.gov/documents/organization/180321.pdf.
68. J. Hofbauer, P. Hermann, and S. Raghavan, Asian defense spending, 2000-2011 (Washington, D.C.: Center for Strategic and International studies, 2012). Available at http://csis.org/files/publication/121005_Berteau_AsianDefenseSpending_Web.pdf.
69. E. A. Friedman and L. O. Gostin, “Pillars for progress on the right to health: Harnessing the potential of human rights through a Framework Convention on Global Health,” Health and Human Rights: An International Journal 14/1 ( 2012), pp. 1-16.
70. Chapman (see note 9).
71. Ajazuddin and Saraf (see note 10).
72. WHO, Médecine traditionnelle (Geneva: WHO, 2002)
73. World Intellectual Property Organization, Guide sur la fixation des savoirs traditionnels (Geneva: WIPO, 2012).
74. T. P. Fan, G. Deal, H. L. Koo, et al., “Future development of global regulations of Chinese herbal products,” Journal of Ethnopharmacology 140 (2012), pp. 568-586.
75. Buyse (see note 11).
76. Zollman and Vickers (see note 16).

